Chemistry > EXAM REVIEW > University of North Carolina, Charlotte - CHEMISTRY 1252Chemistry 1252 S18 Final Exam Review. (All)
University of North Carolina, Charlotte - CHEMISTRY 1252Chemistry 1252 S18 Final Exam Review.
Document Content and Description Below
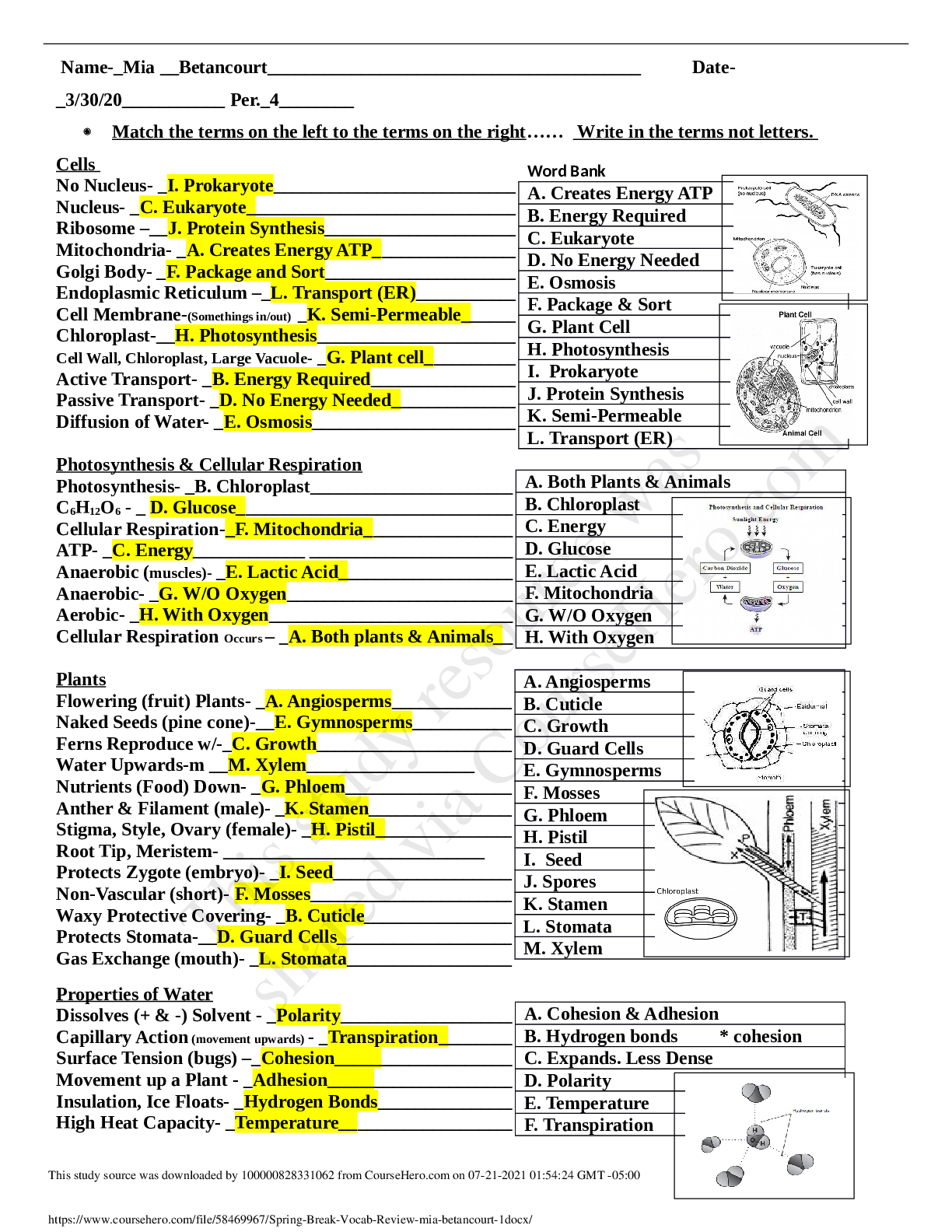
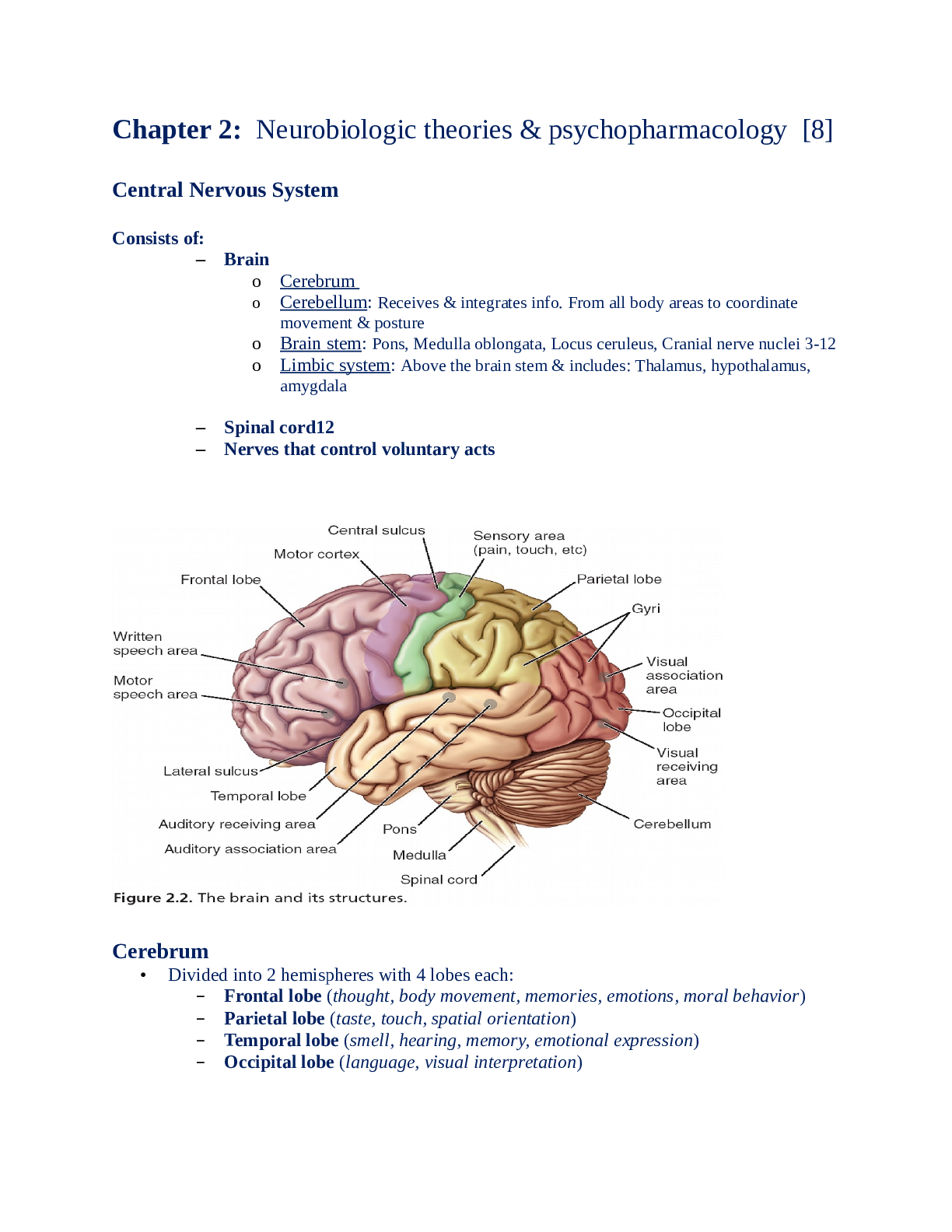
Chemistry 1252 S17 Final Exam Review 1. If 14.0 g of CO2 gas has a volume of 2.3104 mL at 28 C, what is its pressure, in atm? 2. 1.50 atm of wet ammonia gas is dried, then decreased from 78.0... mL to 40.0 mL at constant temperature. What is the final pressure of dry ammonia, in mm Hg, if PH2O = 28.3 mm Hg when the gas was collected? 3. A scuba tank gas mixture contains 3.0 grams of oxygen to every 97.0 grams of helium. What is the partial pressure of oxygen when this mixture is delivered at a total pressure of 8.7 atm?2 4. A 6.100 g sample of an ideal gas in a 2.00 L container is held at a constant pressure of 0.900 atm and a constant temperature of 290.0 K. Calculate the molar mass of the gas. 5. (a)Draw accurate, 3‐D structures for PCl3 and PCl5 including all lone pairs. Draw a crossed arrow to show the molecular dipole for the polar compound. PCl3 PCl5 (b)Which compound would boil at the higher temperature? Justify your answer, using intermolecular forces to explain the differences. 6. Identify the primary intermolecular force experienced by pure samples of (a) through (d). (a) H2S (b) CH3OH (c) H3C-CH2-CH3 (d) He (e) Rank these in order of increasing vapor pressure (at the same temperature). 7. Which of the following molecules can form hydrogen bonds with other molecules of the same kind? Circle your answer(s). [Show More]
Last updated: 1 year ago
Preview 1 out of 10 pages
Instant download

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Mar 11, 2021
Number of pages
10
Written in
Additional information
This document has been written for:
Uploaded
Mar 11, 2021
Downloads
0
Views
52




.png)


.png)