Southern Methodist University - CHEM 3372Exam one chap 13-15
Document Content and Description Below
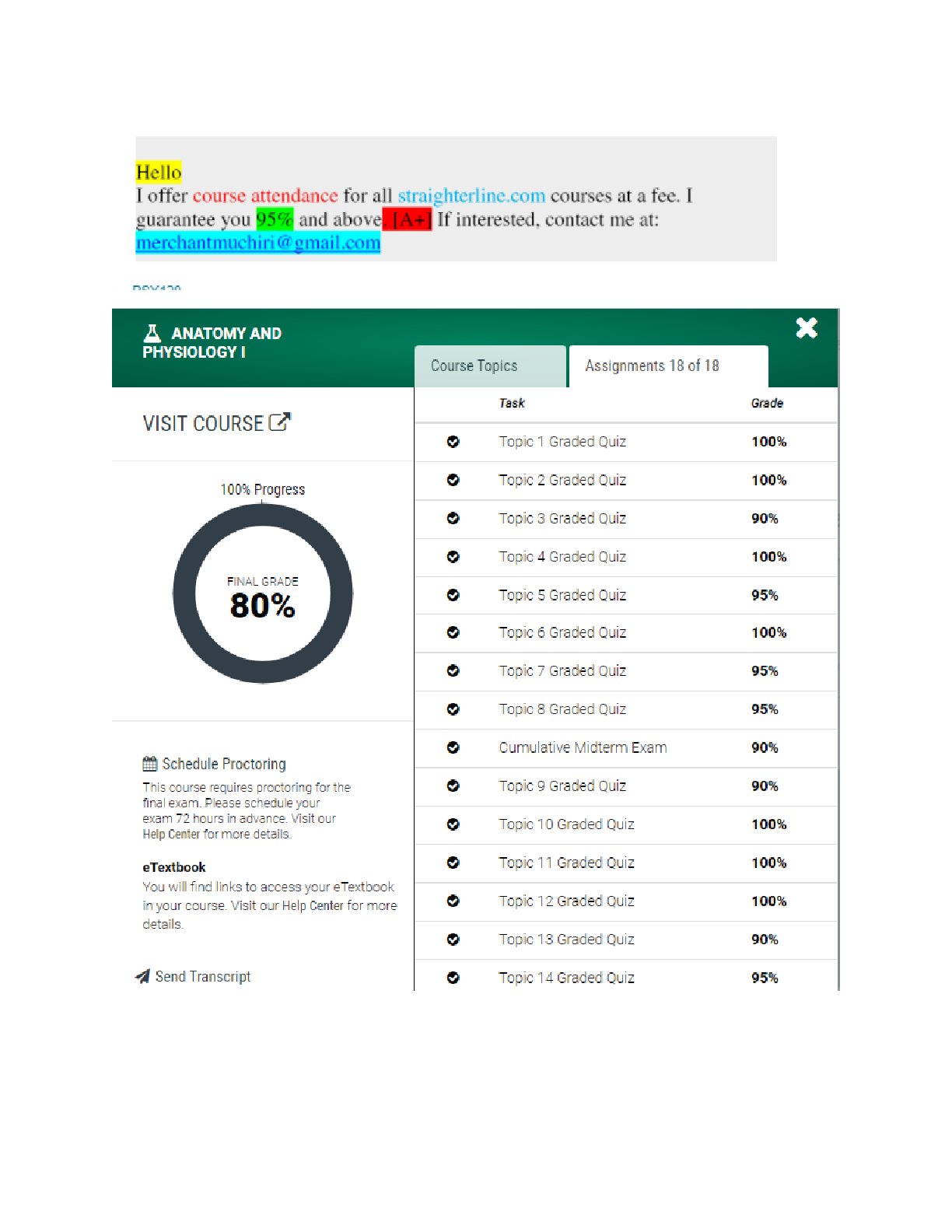
CHEM 3372 Name___________________________________________ Exam #1 February 11, 2014 100 points + 5 extra credit points !!!!Make sure you put your name on three things before turning in the exam!!!... ! 1. This cover sheet 2. The scantron form 3. The free response page2 Multiple choice. Please fill in your answers on the Scantron sheet. 1. Which one of the following statements is not true? (a) The molecular ion peak in a mass spectrum corresponds to the mass of a radical cation. (b) The molecular ion peak for 13CH4 would appear at m/z 17. (c) Infrared radiation possesses lower energy than visible light. (d) Most of the IR absorptions in the 4000–1500 cm–1 region correspond to bending frequencies. 2. Which one of the following statements is not true? (a) The approximate frequency required for 13C nuclei to achieve resonance is greater than that required for 1H nuclei. (b) In NMR spectroscopy, a stronger magnet will require a higher frequency of electromagnetic radiation to achieve resonance. (c) If all the carbon atoms in propane were 13C, you would observe carbon-carbon coupling in the 13C NMR spectrum. (d) For a typical 1H NMR sample, we use CDCl3 rather than CHCl3 as the solvent because the protons in CHCl3 would overpower the signal of the sample being analyzed. 3. In the 1H NMR spectrum of chlorocyclopropane, the signal for Ha would be split into how many peaks? [Show More]
Last updated: 1 year ago
Preview 1 out of 15 pages
Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Reviews( 0 )
$7.00
Document information
Connected school, study & course
About the document
Uploaded On
Mar 14, 2021
Number of pages
15
Written in
Additional information
This document has been written for:
Uploaded
Mar 14, 2021
Downloads
0
Views
26






.png)
.png)
.png)