GIZMOS Nuclear Decay-ALL ANSWERS CORRECT
Document Content and Description Below
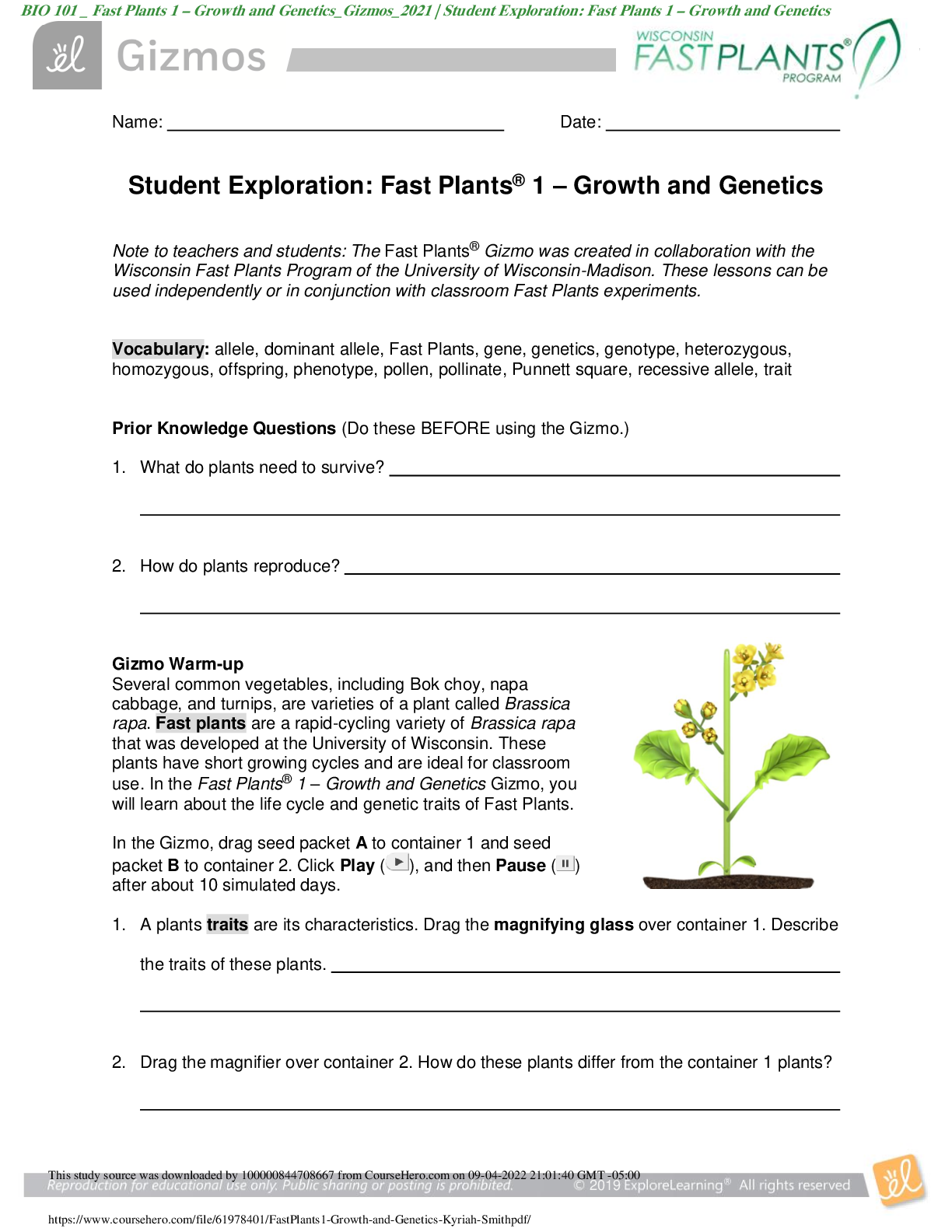
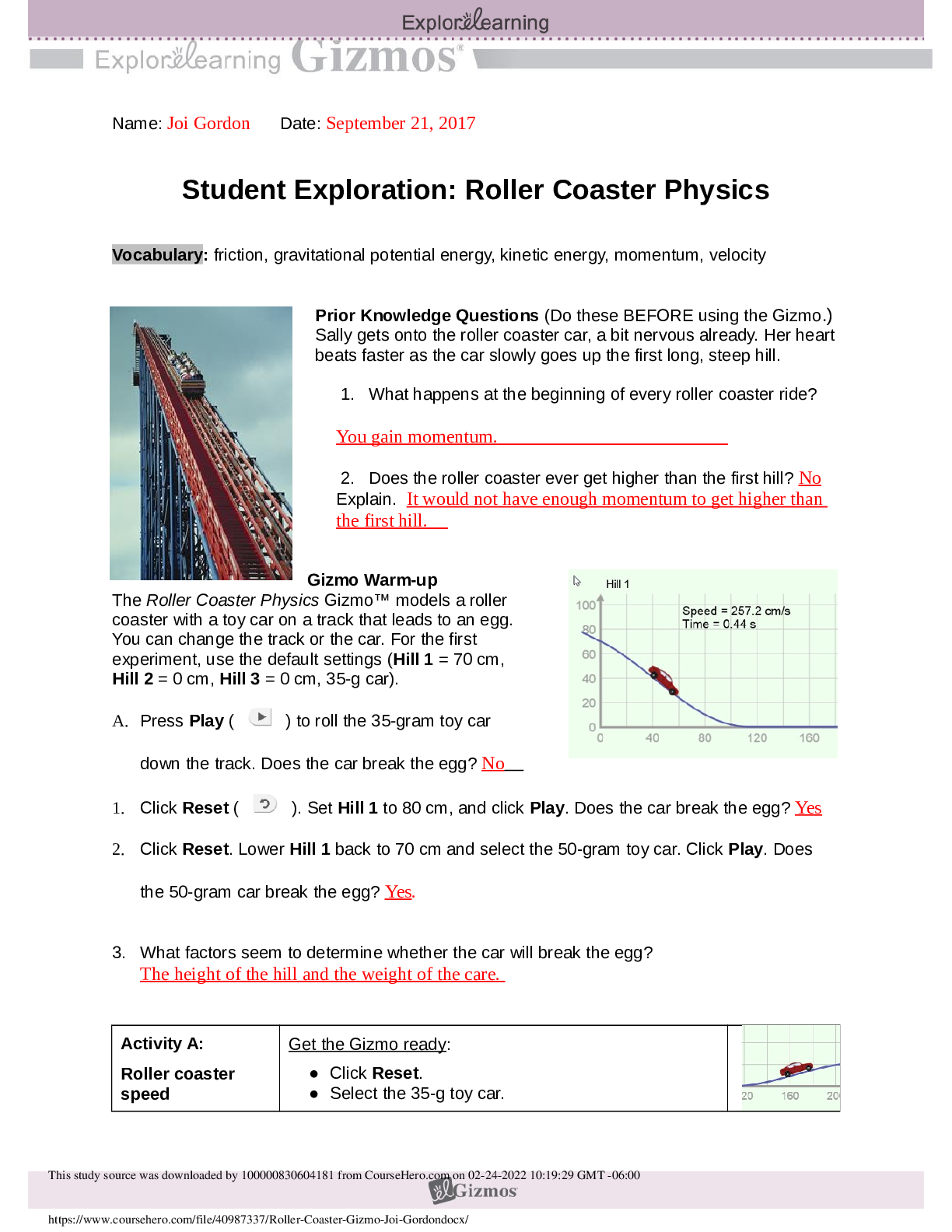
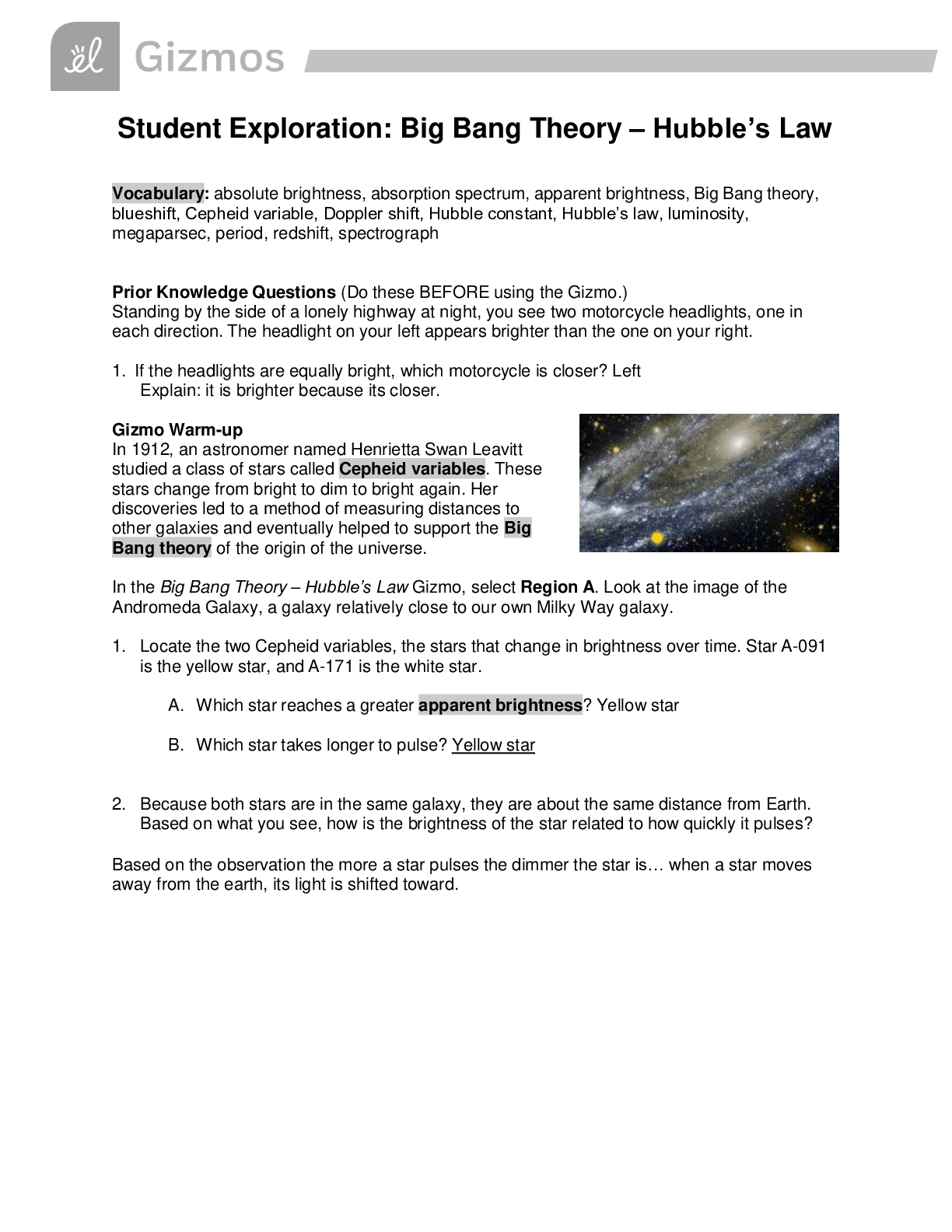
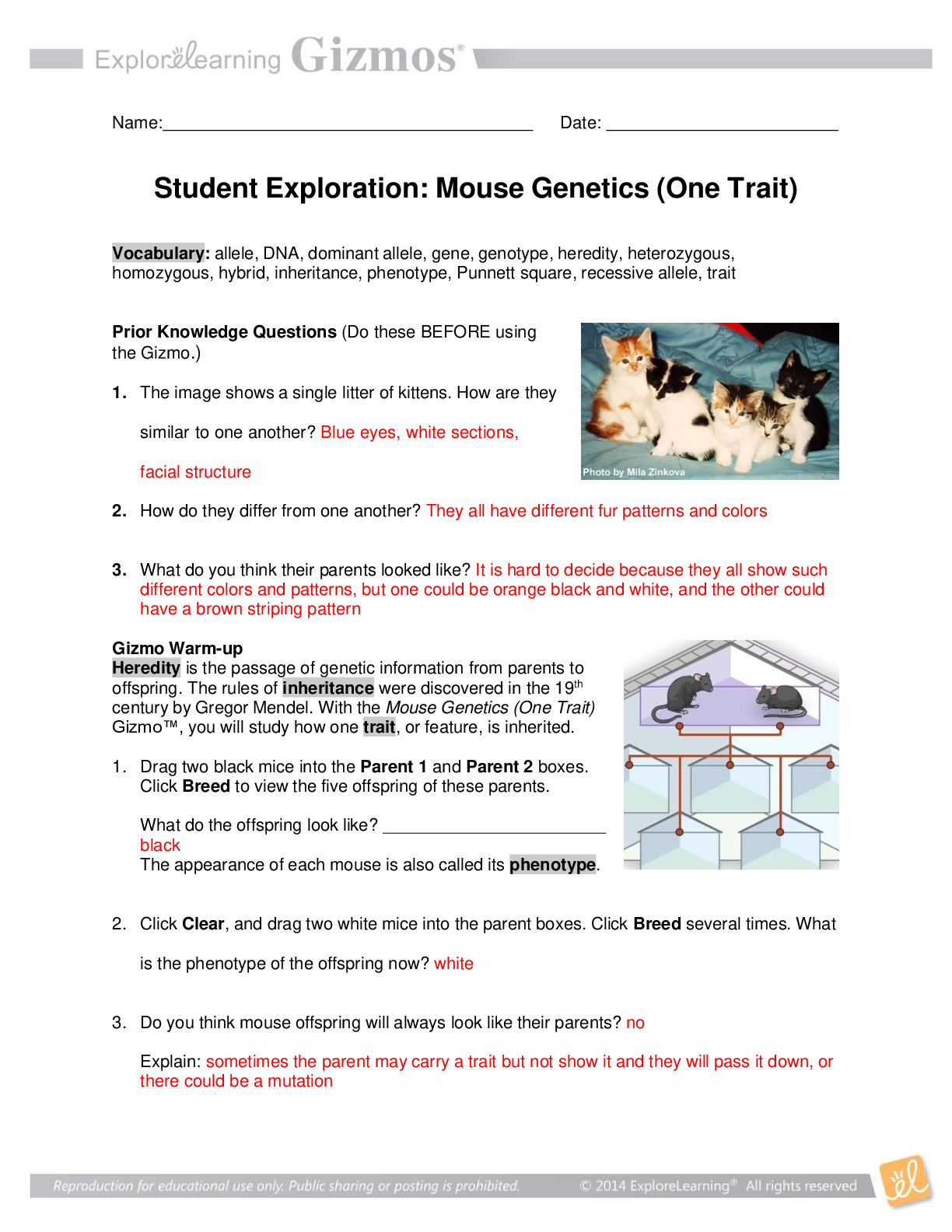
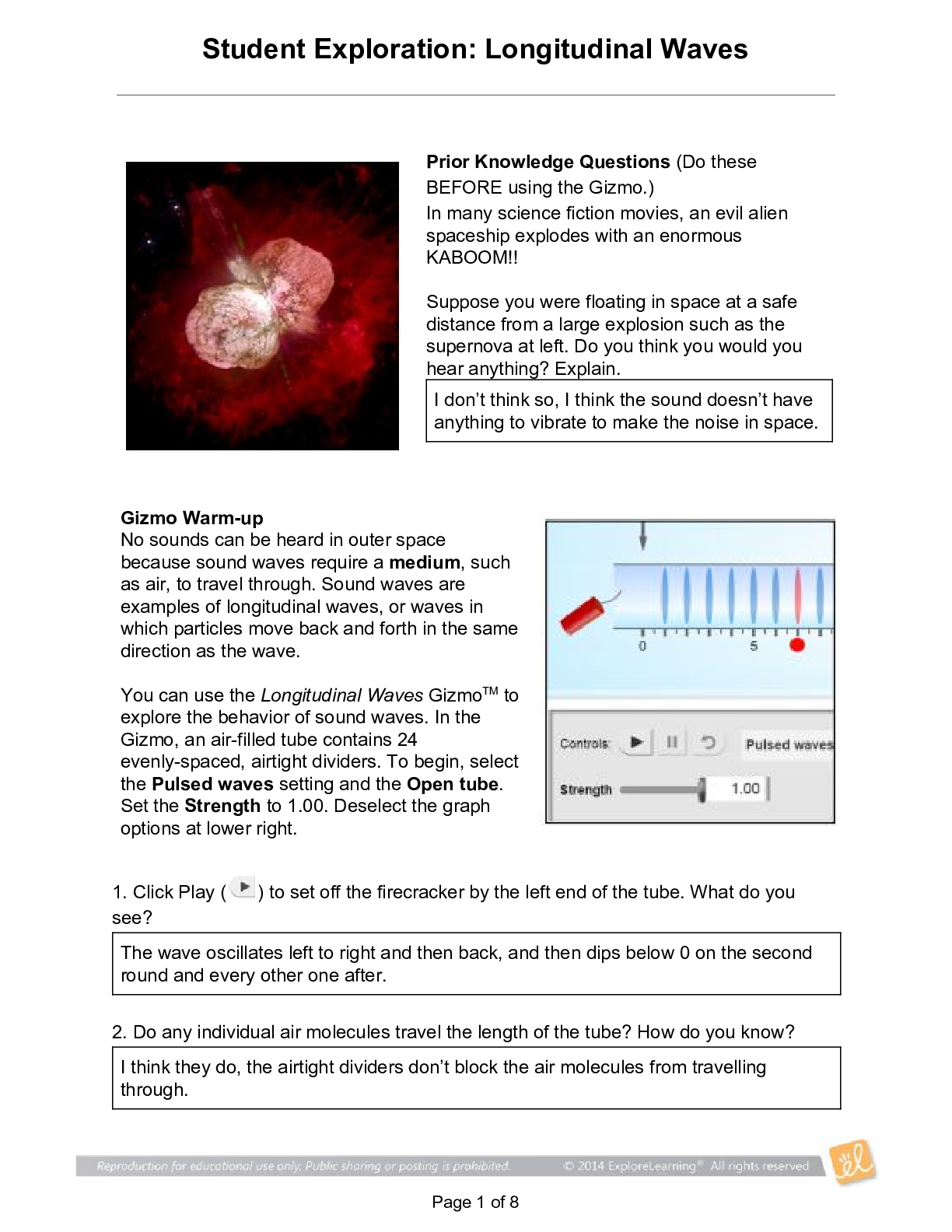
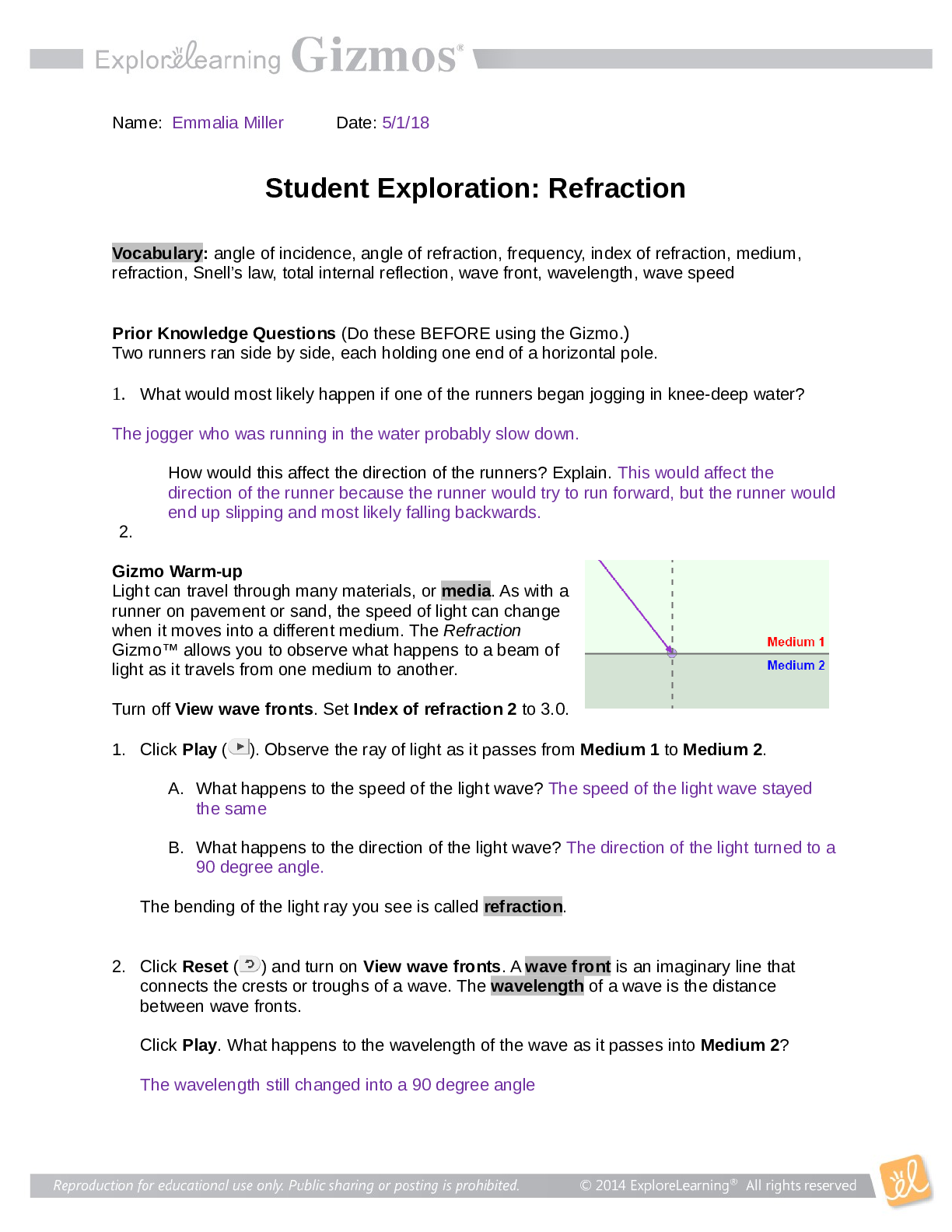
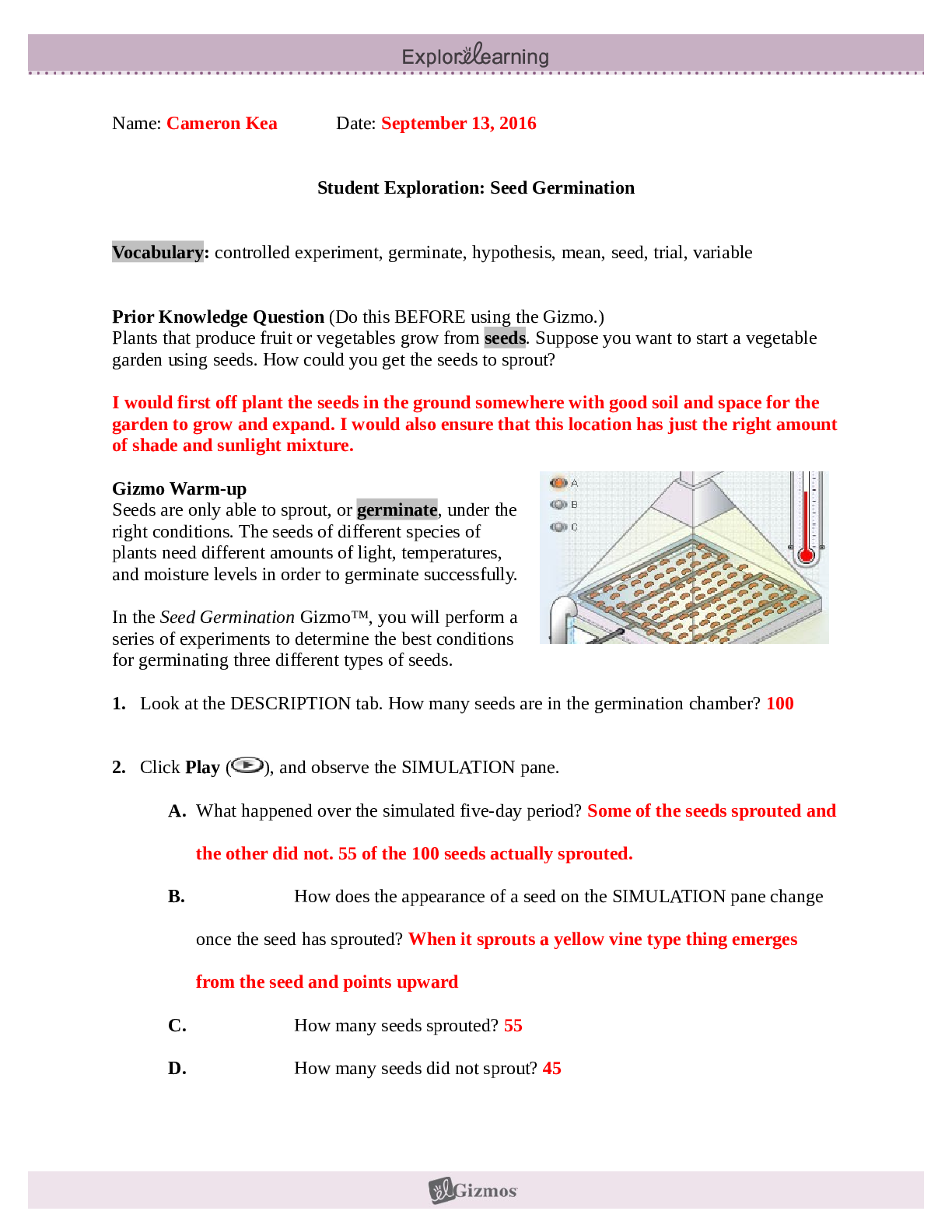
Student Exploration: Nuclear Decay NCVPS Chemistry Fall 2014 Vocabulary: alpha particle, atomic number, beta particle, daughter product, gamma ray, isotope, mass number, nuclear decay, positr... on, radioactive, subatomic particle Prior Knowledge Questions (Do these BEFORE using the Gizmo.) The chart below gives the locations, charges, and approximate masses of three subatomic particles. The approximate mass of each particle is given in universal mass units (u). The mass number of an atom is equal to the sum of protons and neutrons in the nucleus. A helium atom has 2 protons and 2 neutrons. What is the mass number of this atom? 4 The atomic number of an element is equal to the number of protons in each atom of the element. All helium atoms have 2 protons. What is the atomic number of helium? 2 Gizmo Warm-up While most atoms are stable, some are radioactive, which means that they have a tendency to undergo spontaneous nuclear decay. The decay of radioactive atoms generally results in the emission of particles and/or energy. [Show More]
Last updated: 1 year ago
Preview 1 out of 3 pages
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
May 07, 2021
Number of pages
3
Written in
Additional information
This document has been written for:
Uploaded
May 07, 2021
Downloads
0
Views
193

























.png)