Chemistry > Lab Report > CHEM UNIT 7 LAB REPORT UNIT 7 CALORIMETRY EVALUATION (All)
CHEM UNIT 7 LAB REPORT UNIT 7 CALORIMETRY EVALUATION
Document Content and Description Below
1. A 445 g sample of ice at –58oC is heated until its temperature reaches –29oC. Find the change in heat content of the system. Q=C x P x ∆T The temperature change is −29−(−58) =29 Th... e enthalpy change is thus 445g * (29’C) * 2.077 J/g-’C = 271000 Joules = 26.8 kJ 2. A 152 g sample of ice at –37oC is heated until it turns into liquid water at 0oC. Find the change in heat content of the system. Q=C x P x ∆T+ C x m= (152x(0-(-37))x2.10)+ (332x152) = -6.23 x 104 J 3. A 218 g sample of steam at 121oC is cooled to ice at –14oC. Find the change in heat content of the system. Q=C x P x ∆T= 218x(-14-121)x2.10= -6.71 x 104 J 4. If 161 g of water at 85oC is cooled to ice at 0oC, find the change in heat content of the system. Q=C x P x ∆T+ C x m = 161x(0-85)x4.184 + (333 x 161) = 110871 J 5. A 79 g sample of water at 21oC is heated until it becomes steam with a temperature of 143oC. Find the change in heat content of the system. Total Q=(C x P x ∆T)+ (C x m) + (C x m x ∆T)= –2.11 x 105 J 6. If a 348 g sample of steam at 127oC is cooled to 103oC, find the change in heat content of the system. Q=C x P x ∆T= 348x(103-127)x1.87= –1.56 x 104 J 7. In going from ice at –34oC to steam at 138oC, a sample of water absorbs 1.41 x 105 J. Find the mass of the sample. 44.7 g Answers: 1. 2.68 x 104 J 2. 6.23 x 104 J 3. –6.71 x 105 J 4. –1.11 x 105 J 5. 2.11 x 105 J 6. –1.71 x 104 J 7. 44.7 g 8. You find a penny in the snow. How much heat is absorbed by the penny as it warms from the temperature of the snow, which is -8.0oC, to the temperature of your body, 37oC? Assume the penny is pure copper and has a mass of 31.0 g. Use the information from question #14 for the specific heat of copper. 537.07 g 9. Consider the following specific heats of metals. Metal Specific Heat copper 0.385 J/(g • °C) cobalt 0.418 J/(g • °C) chromium 0.447 J/(g • °C) gold 0.129 J/(g • °C) silver 0.237 J/(g • °C) If 100-g samples of each of the metals at 95°C are added to 100 mL of water at 25°C. Which element from the table increase the temperature of the water the greatest amount? Heat released = heat absorbed 100 x c (95- T) = 100 x 4.18 x (T-25) T= (9500 c + 10450)/(100c +418) Since temperature will be lowest when the c is lower So the correct answer is GOLD because it has the lowest specific heat And it will increase the water temperature 10. The specific heat capacity of methane gas is 2.20 J/g-°C. How many joules of heat are needed to raise the temperature of 5.00 g of methane from 36.0°C to 75.0°C? E = 2.20 J/gK x 5g x 39K = 429 11. Which of the following processes is exothermic? A) liquid water condensing from steam B) the melting of ice C) the chemical reaction in a "cold pack" often used to treat injuries D) sweat evaporating from skin E) None of the above are exothermic. Option D: sweat evaporating from skin [Show More]
Last updated: 1 year ago
Preview 1 out of 3 pages

Reviews( 0 )
Recommended For You
Computer Science> Lab Report > COMP 230 Week 7 Lab Report: VBScript Database Query Lab Report (GRADED A) (All)

COMP 230 Week 7 Lab Report: VBScript Database Query Lab Report (GRADED A)
COMP 230 Week 7 Lab Report: VBScript Database Query Lab Report (GRADED A)
By A+ Solutions , Uploaded: May 05, 2022
$10.5
Chemistry> Lab Report > University of the Fraser Valley - CHEM MISCFernandez, Tatiana - Unit 7 Lab Report Unit 7 - Calorimetry Evaluation (All)

University of the Fraser Valley - CHEM MISCFernandez, Tatiana - Unit 7 Lab Report Unit 7 - Calorimetry Evaluation
Unit 7 - Calorimetry Evaluation 1. A 445 g sample of ice at –58oC is heated until its temperature reaches –29oC. Find the change in heat content of the system. Q = cm∆T Q = ".$% ' ( ∙ ℃ X 445 g...
By Muchiri , Uploaded: Apr 05, 2021
$8.5
Physics> Lab Report > PHY 2048L Experiment 4 Lab Report: Force Table, Florida Atlantic University (All)

PHY 2048L Experiment 4 Lab Report: Force Table, Florida Atlantic University
PHY2048L 007: Experiment 4: Force Table February 14, 2020 Purpose The purpose of this experiment is to add a set of vectors using three different methods (graphical, analytical, and experimental)...
By Kirsch , Uploaded: Oct 01, 2020
$9
Chemistry> Lab Report > CHEM 162 Experiment #5 Spectrochemical Series (All)

CHEM 162 Experiment #5 Spectrochemical Series
CHEM 162 Experiment #5: Spectrochemical Series Goals of this lab: • Produce a copper coordination compound and evaluate the yield • Calculate d-orbital splitting energies from spectral data in nano...
By Muchiri , Uploaded: May 20, 2021
$9
Environmental Science> Lab Report > ENVR 1401Lab 11 toxicology experiment and lab report Collin College - (All)

ENVR 1401Lab 11 toxicology experiment and lab report Collin College -
Lab 11 Toxicology Experiment and Lab report assignment Project deliverables: At the end of this project you will need to upload a copy of your lab assignment and report (see guidelines below). You...
By Muchiri , Uploaded: Apr 06, 2021
$10
Biology> Lab Report > BIO 208 Reproductive System, and Development, Growth, and Aging Lab Practical 2. All Correct Answers Indicated. (All)
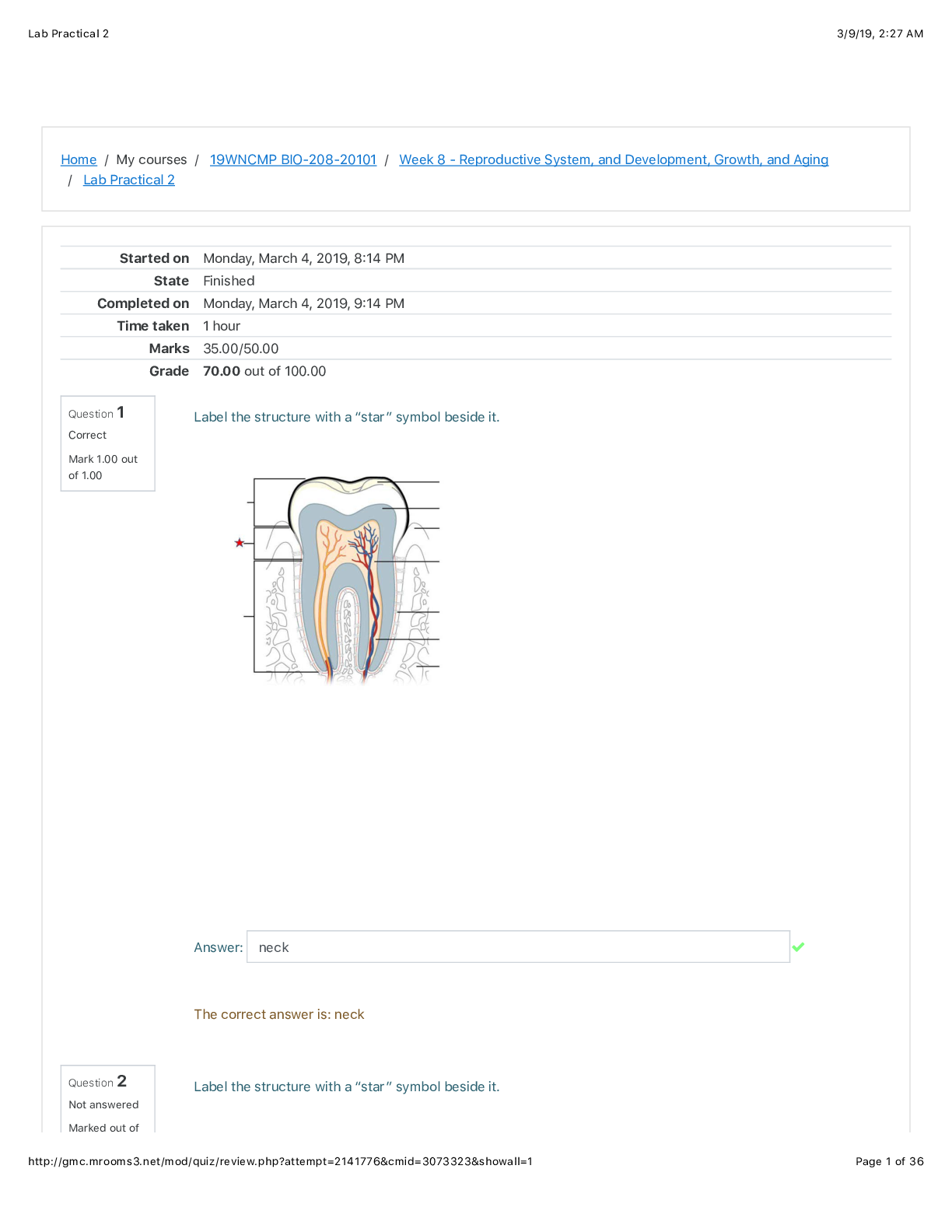
BIO 208 Reproductive System, and Development, Growth, and Aging Lab Practical 2. All Correct Answers Indicated.
Lab Practical Home / My courses / Question 1 Correct Mark 1.00 out of 1.00 Question 2 Not answered Marked out of Started on Monday, March 4, 2019, 8Q14 PM State Finished Completed on M...
By Kirsch , Uploaded: Nov 27, 2020
$10
Physics> Lab Report > PHY 110 Lab 7 - Forces in Every Life. University of New England (All)

PHY 110 Lab 7 - Forces in Every Life. University of New England
Forces in Everyday Life (Week 7) Model or Topic Explored: You will create a demonstration of either the free particle (sum of the forces is equal to zero) or constant force particle (sum of the forc...
By Cheryshev , Uploaded: Jul 02, 2021
$8.5
*NURSING> Lab Report > HPE ProLiant Questions and Answers, Lab Reports for Mathematics (All)

HPE ProLiant Questions and Answers, Lab Reports for Mathematics
HPE ProLiant Questions and Answers, Lab Reports for Mathematics 3.8 (14) Refer to the exhibit of an internal view of the HPE ProLiant WS460c Graphics Server Blade.- HPE Smart Array P244br Controlle...
By WINNIE MUMBI , Uploaded: Nov 12, 2023
$21
Chemistry> Lab Report > Unit 2 assignment 2 - Lab Report on Study of Cooling curves (All)

Unit 2 assignment 2 - Lab Report on Study of Cooling curves
Unit 2 assignment 2 - Lab Report on Study of Cooling curves
By ACADEMICTUTORIAL , Uploaded: Dec 22, 2021
$3
Psychology> Lab Report > PSYC 300 The Facial Recognition Mini-Lab Report (All)

PSYC 300 The Facial Recognition Mini-Lab Report
PSYC 300 The Facial Recognition Mini-Lab Report
By ACADEMICTUTORIAL , Uploaded: Mar 30, 2023
$3.5
Document information
Connected school, study & course
About the document
Uploaded On
Jun 10, 2021
Number of pages
3
Written in
Additional information
This document has been written for:
Uploaded
Jun 10, 2021
Downloads
1
Views
459






