*NURSING > EXAM > OCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam (All)
OCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark Scheme
Document Content and Description Below
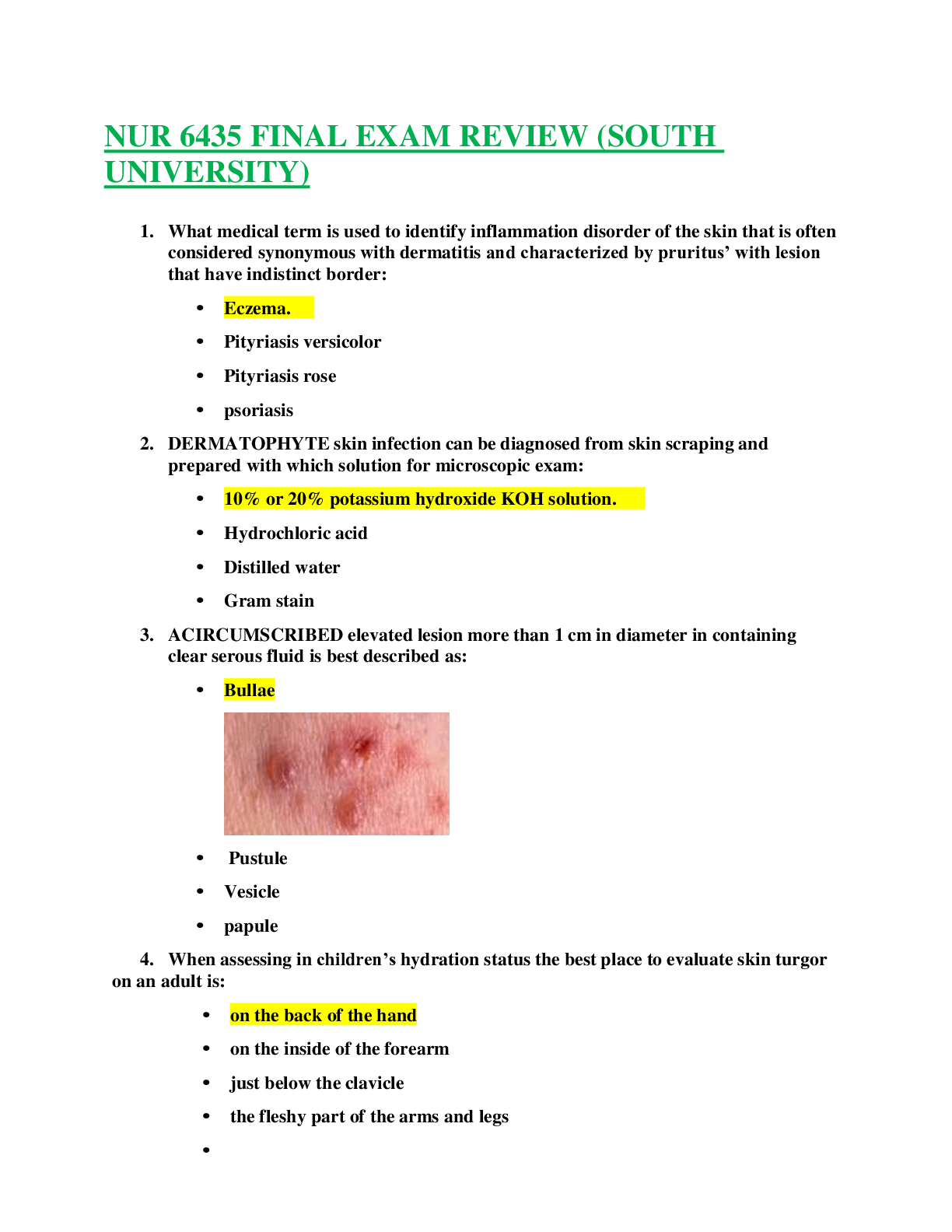
OCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark SchemeOCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an o... rganic solid Practice Exam Questions and Mark SchemeOCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark SchemeOCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark SchemeOCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark SchemeOCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark SchemeOCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark SchemeDevelopment of practical skills in chemistry. A solid organic compound can be purified by recrystallisation. Which statement(s) about recrystallisation is/are true? 1 The organic compound is more soluble in hot solvent. 2 The hot solution is cooled before the purified organic compound is collected. 3 The melting point of the purified organic compound is lower than the impure compound. A 1, 2 and 3 B Only 1 and 2 C Only 2 and 3 D Only 1 Your answer [1] 2. When are insoluble impurities removed during recrystallisation? A when the hot solution is filtered B as the solution cools C when the crystals are filtered off D when the crystals are washed Your answer [1] OCR A Level Chemistry A (H432) Chemistry B (H433) PAG 6: Synthesis of an organic solid Practice Exam Questions and Mark Scheme 2(a). Benzoic acid, C6H5COOH, is added to some foods as a preservative. A student prepares benzoic acid as outlined below. Step 1 The student mixes 4.00 cm3 of phenylmethanol, C6H5CH2OH, (density = 1.04 g cm−3) with sodium carbonate and aqueous potassium manganate(VII), as an oxidising agent. [Show More]
Last updated: 1 year ago
Preview 1 out of 17 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Reviews( 0 )
$14.00
Document information
Connected school, study & course
About the document
Uploaded On
Jul 12, 2021
Number of pages
17
Written in
Additional information
This document has been written for:
Uploaded
Jul 12, 2021
Downloads
0
Views
46






















.png)


