Portage Learning Chem 210 Module 2 Exam-2022
Document Content and Description Below
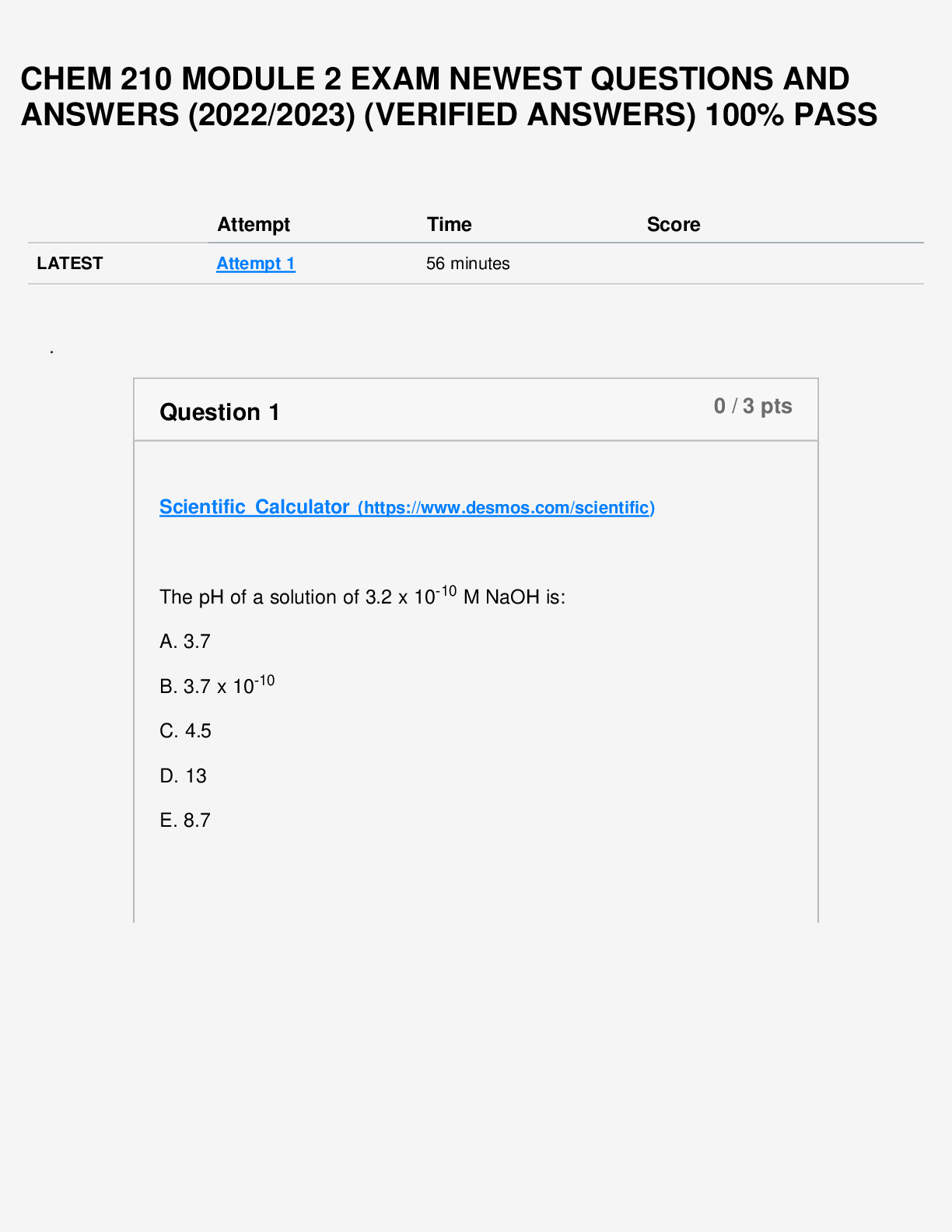
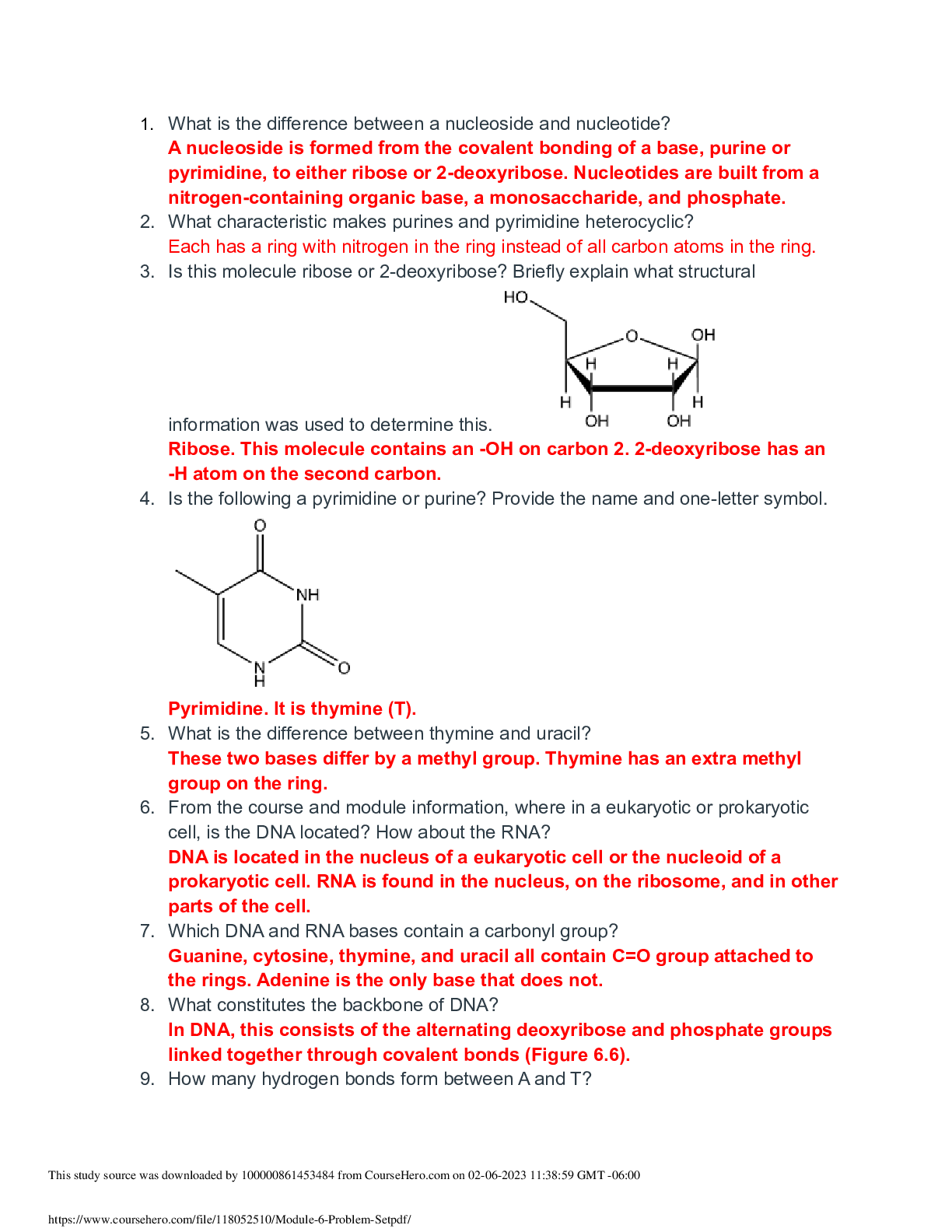
Question 1 3 / 3 pts Scientific Calculator The pH of a solution of 8.9 x 10-12 M NaOH is: A. 8.9 B. 8.9 x 10-12 C. 4.8 D. 11 E. 2.9 A B C D Correct! E [Kw=[H+][OH-] = 1.0 x 10-14; ... [H+][8.9 x 10-12] = 1.0 x 10-14 ;[H+]=890; pH=-log[890]=2.9] Question 2 3 / 3 pts Water is a unique molecule. Which of the following contributes to water’s unique place Portage Learning Chem 210 Module 2 Exam in the chemical world? The geometry of the molecule The polarity of the O-H bonds The ability of water molecules to hydrogen bond The bond angle of water All of the answers are correct ALL OF THE ANSWERS ARE CORRECT What factor contributes to the bent shape of a water molecule? The dipole arrows in a water molecule The unshared electron pairs on the oxygen atom The electronegativity difference between hydrogen and oxygen The unequal electron sharing between hydrogen and oxygen None of the answers are correct THE UNSHARED ELECTRON PAIRS ON THE OXYGEN ATOM Question 4 3 / 3 pts Which diagrams correctly represent a hydrogen bond? A and B A and D B and C C and D A and C A AND C BOTH SHOW A PROTON DONOR AND ACCEPTOR Question 5 3 / 3 pts Scientific Calculator The H+ concentration of a solution is 5.6 x 10-5 What is the pH? -4.25 Correct! 4.25 [ [H+]=5.6 x 10-5; pH=-log[5.6 x 10-5]=4.25] 5.65 -9.75 9.75 4.25 [ [H+]=5.6 x 10-5; pH=-log[5.6 x 10-5]=4.25] If the Ka of an acid is 2.67 x 10-5, what is the pKa? 7.45 12.25 5.26 2.67 4.57 4.57 [pKa = -log(2.67 x 10-5) = 4.57] Question 7 3 / 3 pts List the acids in INCREASING order of strength (weakest to strongest): Nitrous acid (Ka = 4.0 x 10-4); acetic acid (Ka = 1.7 x 10-5); phosphoric acid (Ka = 7.3 x 10-3) Nitrous acid, acetic acid, phosphoric acid Acetic acid, nitrous acid, phosphoric acid Phosphoric acid, nitrous acid, acetic acid Acetic acid, phosphoric acid, nitrous acid None of the above ACETIC ACID, NITROUS ACID, PHOSPHORIC ACID Question 8 3 / 3 pts Elemental bromine (Br2) is a liquid at room temperature. What is the dominant attractive force that exists between Br2 molecules in the liquid? Halogen forces Dipole-dipole forces Hydrogen bonds Covalent bonds London forces LONDON FORCES Which one of the following molecules can form a hydrogen bond? A. HBr B. HCl C. NH3 D. CH4 E. H2 C. NH3 Question 10 3 / 3 pts A hydrogen bond can form between a hydrogen atom bonded to nitrogen, N–H, and ________. C O Cl P Br O Question 11 3 / 3 pts In a bond between P and Cl, there exists a difference in electronegativity resulting in partial positive and partial negative charges. If a dipole arrow were added to this structure, which way would the arrow point? The arrow points from Br toward N The arrow points from N toward Br Two arrows; one pointed in each direction There would not be a dipole arrow in this case. None of the above THE ARROW POINTS FROM N TOWARD BR Question 12 3 / 3 pts The following reaction occurs in glues purchased from hardware stores. Customers use this product to glue plastic together. Epoxy (l) + hardener (l) ⟶ ⟶ Glue (s) + heat given of What is the sign (+ or -) of the change in Δ ΔG and Δ ΔH? A) ΔG is (+) and ΔH is (-) B) ΔG is (-) and ΔH is (-) C) ΔG is (-) and ΔH is (+) D) ΔG is (+) and ΔH is (+) E) None of the above B) ΔG is (-) and ΔH is (-) Question 13 3 / 3 pts Which of the following is the free-energy change of a reaction? A) G B) ΔS C) ΔH D) ΔG E) All of the above D) ΔG Question 14 3 / 3 pts Which of the following is the change in entropy of a reaction? A) ΔG B) ΔE [Show More]
Last updated: 1 year ago
Preview 1 out of 17 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Jul 12, 2022
Number of pages
17
Written in
Additional information
This document has been written for:
Uploaded
Jul 12, 2022
Downloads
0
Views
75