Limiting Reactants Gizmo
Document Content and Description Below
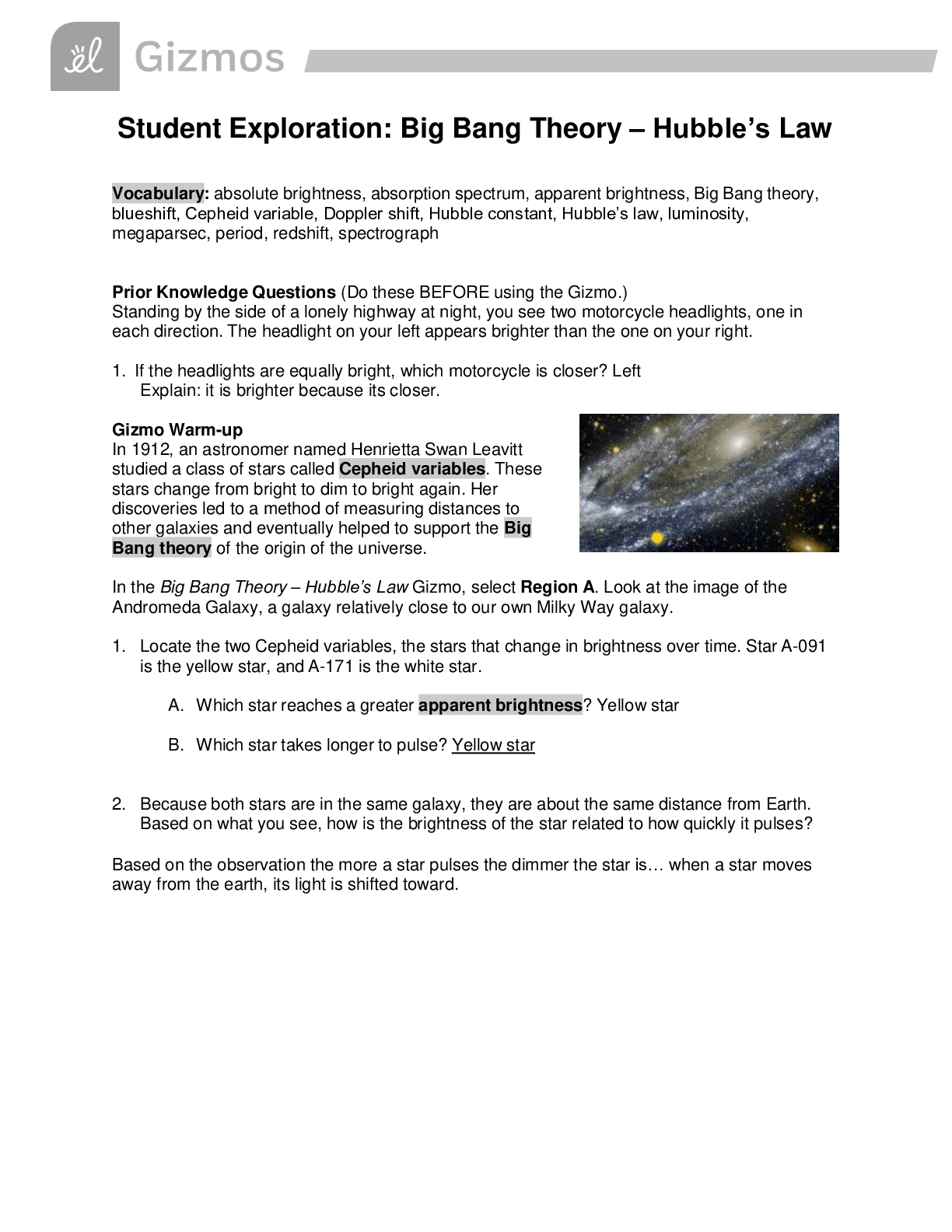
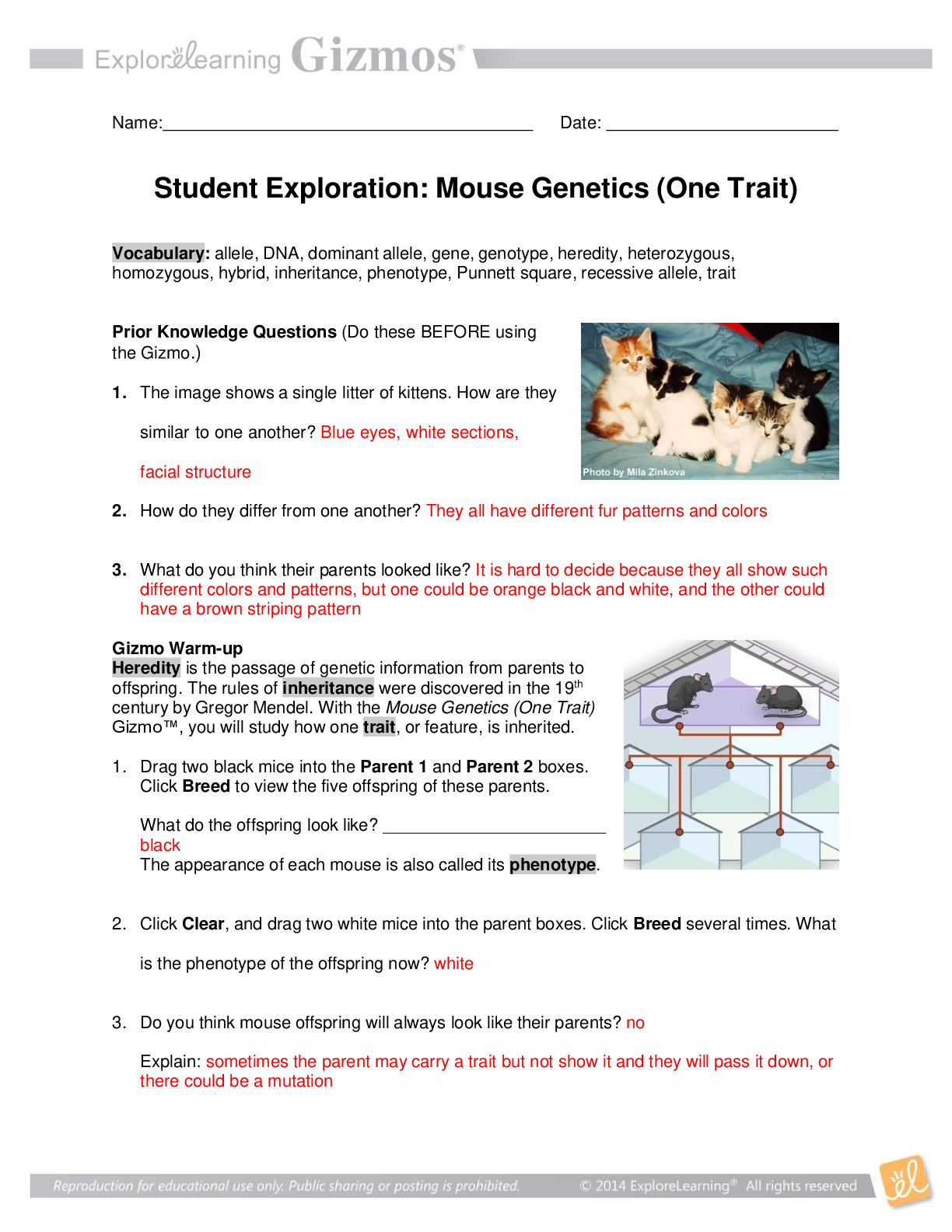
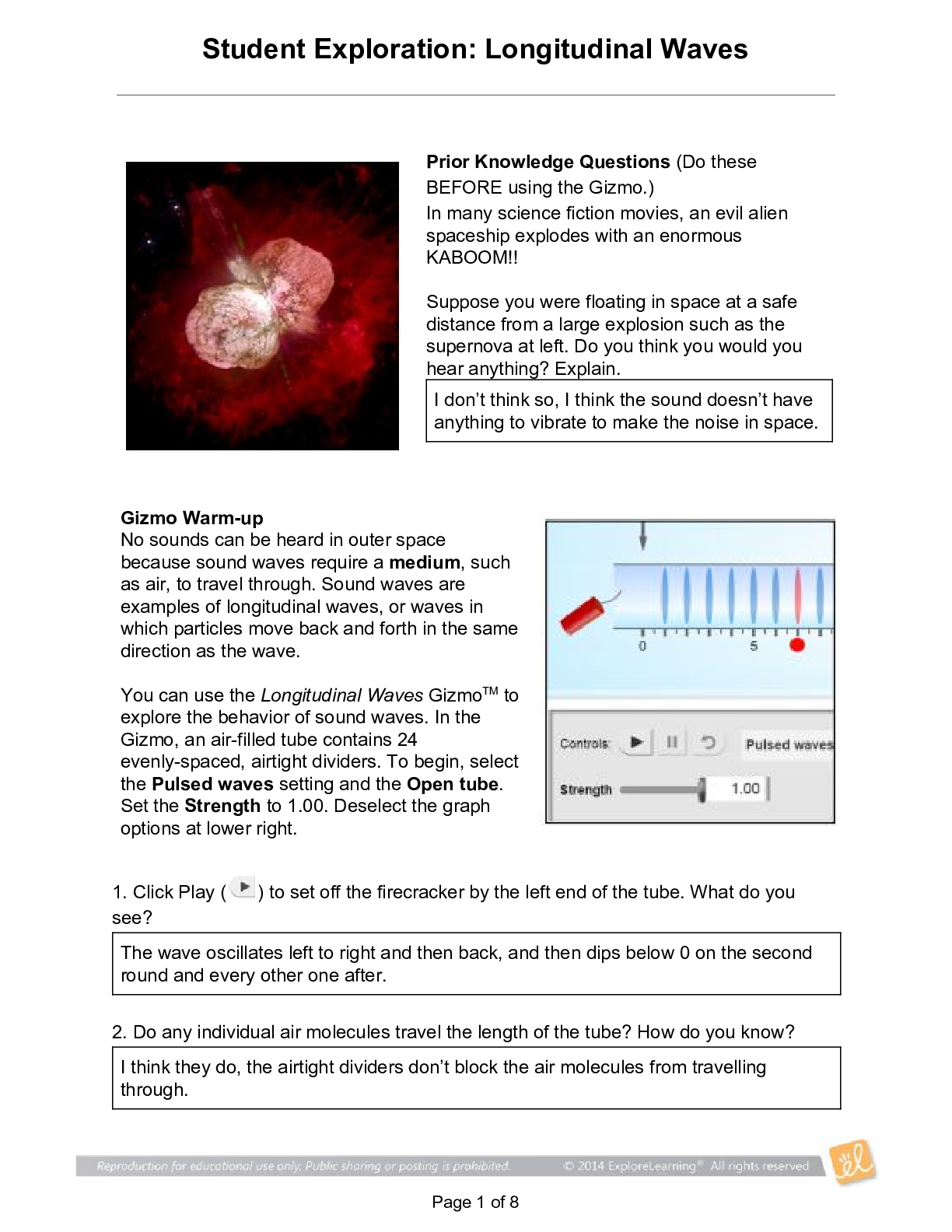
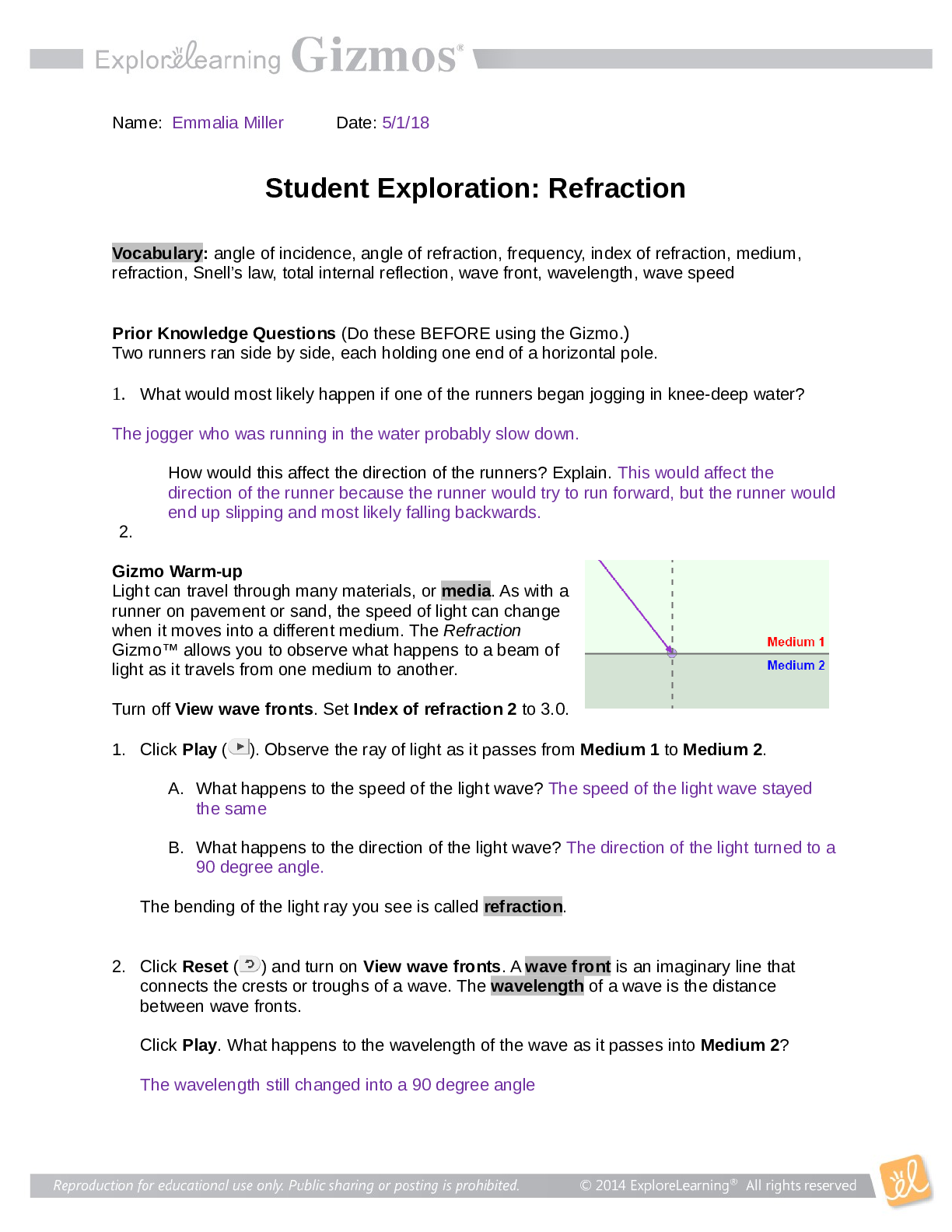
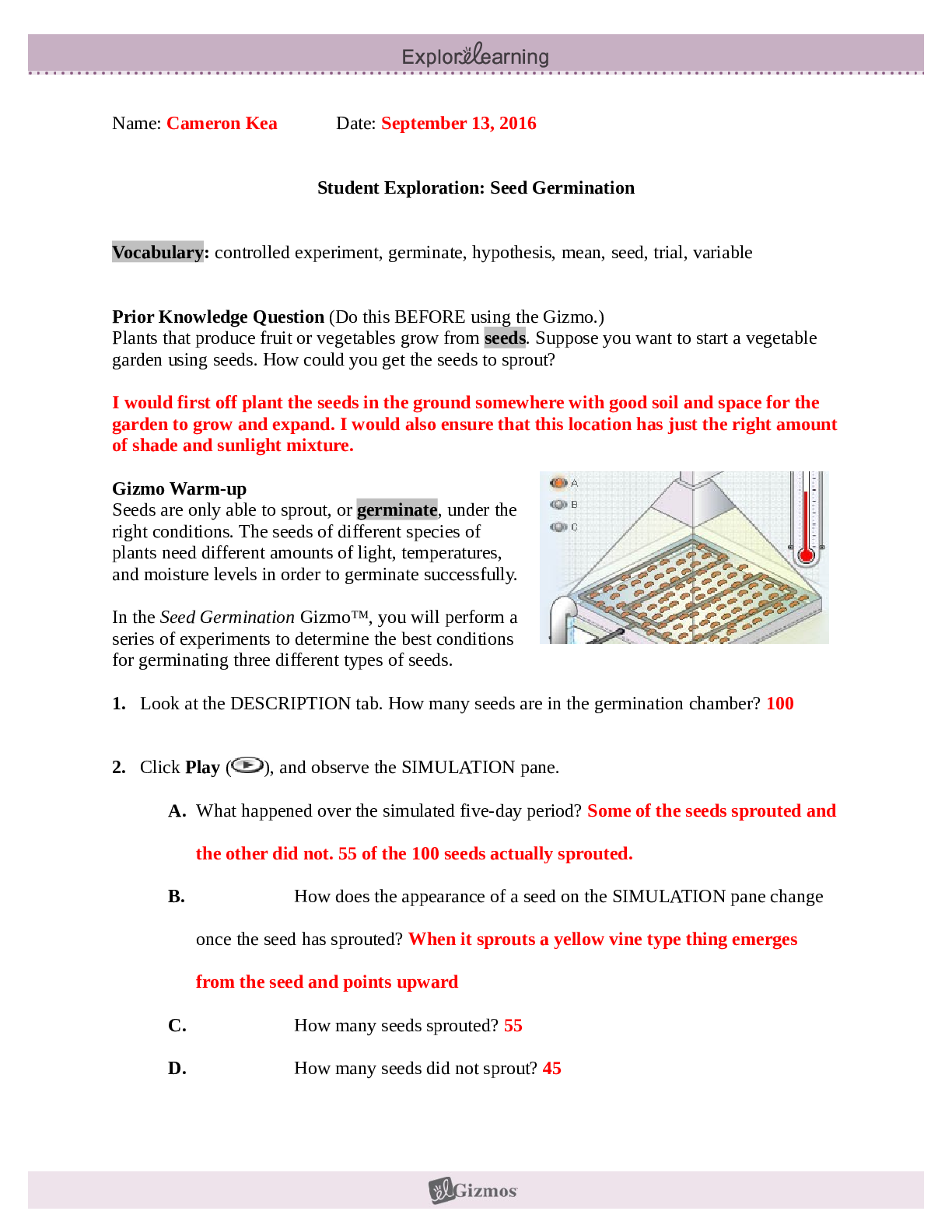
Student Exploration: Limiting Reactants Vocab - Chemical equation – a symbolic representation of a chemical reaction. - Chemical formula – a symbolic representation of an element or compound. ... - Chemical reaction – a process in which one or more substances are transformed into others. - Limiting reactant – the reactant in a chemical reaction that limits the amount of product that is able to form. - Molecule – a stable particle made of two or more atoms. - Product – a substance that is formed in a chemical reaction. - Reactant – a substance that takes part in a chemical reaction. - Subscript – a number in a chemical formula representing the number of atoms of a particular element in one molecule of the compound. Prior Knowledge Questions 1. How many total hot dogs can you make? 30 2. Which ingredient limited the number of hot dogs you could make? buns 3. Which ingredient will you have leftovers of? Hot dogs Gizmo Warm-up 1. Use the sliders to set the number of O2 molecules and H2 molecules to two. a. How many hydrogen molecules (H2) are there? 2 Hydrogen atoms? 4 [Show More]
Last updated: 1 year ago
Preview 1 out of 2 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Sep 29, 2022
Number of pages
2
Written in
Additional information
This document has been written for:
Uploaded
Sep 29, 2022
Downloads
0
Views
115















.png)

.png)

