Engineering > QUESTIONS & ANSWERS > BIOE 349 Metabolic engineering - Lehigh University _ Metabolic Engineering: Homework 7 Solutions – (All)
BIOE 349 Metabolic engineering - Lehigh University _ Metabolic Engineering: Homework 7 Solutions – 9 points
Document Content and Description Below
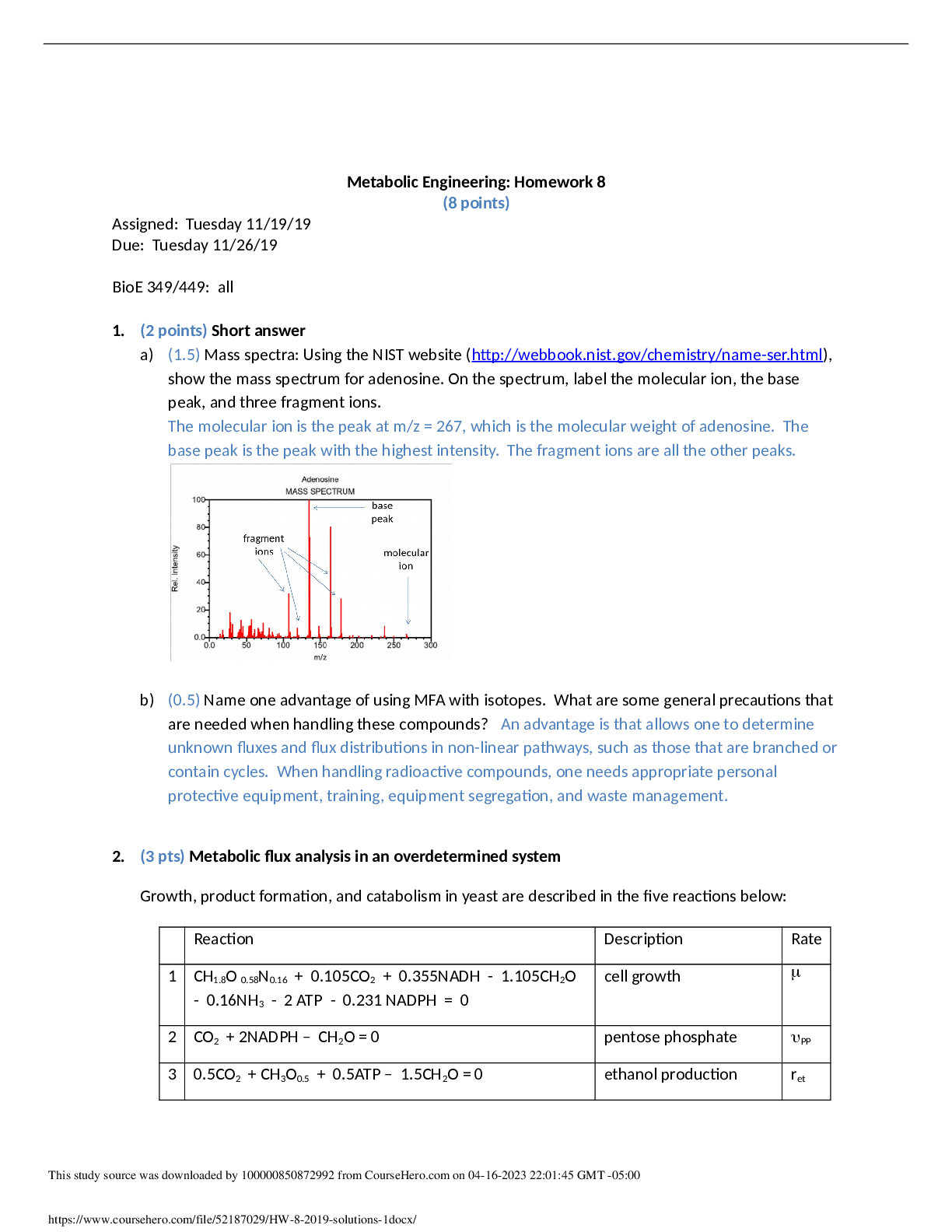
Metabolic Engineering: Homework 7 Solutions – 9 points 1. (2pts) Short answer (a) (0.5pt) A reaction has GR > 0. Is energy consumed or produced? Express GR in terms of Gcat. When ... GR > 0, energy is consumed. GR = - GCAT (b) (0.5pt) In class, we discussed that the theoretical value of the P/O ratio typically greater than the actual (or operational) value due since there are other physiological mechanisms, besides ATP generation via oxidative phosphorylation, that utilize proton gradients. Provide an example of one such mechanism. (c) (1) In "Stoichiometric balance based macrokinetic model for Penicillium chrysogenum in fed-batch fermentation" Yuan, et. al. simplify the penicillin pathway into a lumped model. 1 Name three examples in this pathway of reactions that are lumped together. 2 The specific rate of product formation (qp) is described by the Luedeking-Peret model, qp = . Is this a catabolic or an anabolic model for product formation? How do you know? (Compare this to the qp models presented in Ch 9 of Smolke.) 3 Why did the authors include the regulator model to describe product formation? . 4 Was the biomass in the system assumed to be homogeneous, or not? Explain. Does this mean that the system is structured or unstructured? 2. (3 points) Stoichiometric models: Anaerobic biosynthesis & catabolism in yeast The biosynthetic and catabolic reactions in yeast can be expressed using the five reactions below: Reaction Description Rate 1 CH1.8O 0.58N0.16 + 0.105CO2 + 0.355NADH - 1.105CH2O - 0.16NH3 - YXATPATP - 0.231 NADPH = 0 cell growth 2 CO2 + 2NADPH – CH2O = 0 pentose phosphate PP 3 0.5CO2 + CH3O0.5 + 0.5ATP – 1.5CH2O = 0 ethanol production ret 4 CH8/3O - 0.333NADH - 0.333ATP - CH2O = 0 glycerol production rgly 5 -ATP = 0 ATP consumption for maintenance mATP Notes: glucose = CH2O; ethanol = CH3O0.5; glycerol= CH8/3O. The rate vector v for the system above can be expressed as: (a) (0.25) Is this a detailed stoichiometric model or a grouped (aka "lumped") stoichiometric model? What is the basis for your conclusion? (b) (0.25) Express the 5 reactions in matrix notation, i.e. AS + BP + Xmacro + GXmet = 0 Neglect ammonia and assume that your only substrate is glucose. Products: carbon dioxide, ethanol, glycerol Intracellular products: ATP, NADH, NADPH (c) (0.5)Express the rates in matrix notation. i.e. rS=-ATv, rP=BTv, rmacro=Tv, rmet=GTv. (d) (0.5)What rates are approximately equal to zero, based upon the pseudo-steady-state approximation? Using this assumption, express rgly and PP in terms of and express ret in terms of , YXATP, and mATP. [Show More]
Last updated: 1 year ago
Preview 1 out of 8 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Reviews( 0 )
$9.50
Document information
Connected school, study & course
About the document
Uploaded On
Apr 13, 2023
Number of pages
8
Written in
Additional information
This document has been written for:
Uploaded
Apr 13, 2023
Downloads
0
Views
35