Chemistry > QUESTIONS & ANSWERS > CHEMISTRY_ Using the expression Ka=[H+][A−] / [HA], explain how to determine which solution has th (All)
CHEMISTRY_ Using the expression Ka=[H+][A−] / [HA], explain how to determine which solution has the lower pH , 0.10 M HF(aq) or 0.10 M HC2H3O2(aq). Do not perform any numerical calculations.
Document Content and Description Below
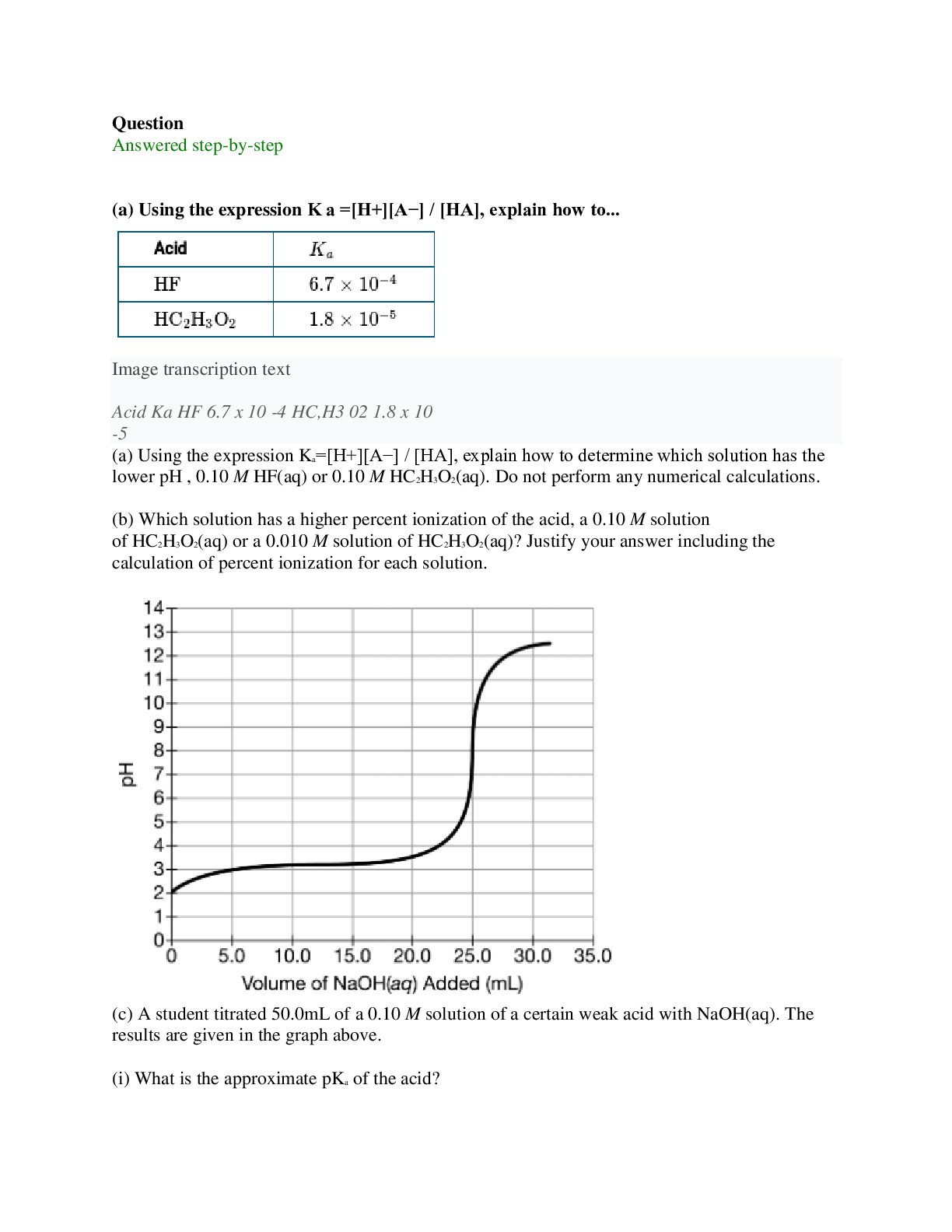
(a) Using the expression Ka=[H+][A−] / [HA], explain how to determine which solution has the lower pH , 0.10 M HF(aq) or 0.10 M HC2H3O2(aq). Do not perform any numerical calculations. (b) Whi... ch solution has a higher percent ionization of the acid, a 0.10 M solution of HC2H3O2(aq) or a 0.010 M solution of HC2H3O2(aq)? Justify your answer including the calculation of percent ionization for each solution. (c) A student titrated 50.0mL of a 0.10 M solution of a certain weak acid with NaOH(aq). The results are given in the graph above. (i) What is the approximate pKa of the acid? (ii) What specific value from the graph is needed, in addition to the information in part (c) above, to calculate the molar concentration of the NaOH(aq)? (iii) Identify a pH value between 2.5 and 7.5 at which the concentration of the weak acid being titrated is less than the concentration of its conjugate base. (d) In another experiment, the student titrated 50.0mL of 0.100 M HC2H3O2 with 0.100 M NaOH(aq). Calculate the pH of the solution at the equivalence point. [Show More]
Last updated: 1 year ago
Preview 1 out of 3 pages
Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Reviews( 0 )
$9.50
Document information
Connected school, study & course
About the document
Uploaded On
Apr 20, 2023
Number of pages
3
Written in
Additional information
This document has been written for:
Uploaded
Apr 20, 2023
Downloads
0
Views
61







.png)

