Average atomic mass (Well structured Answers in Bold)
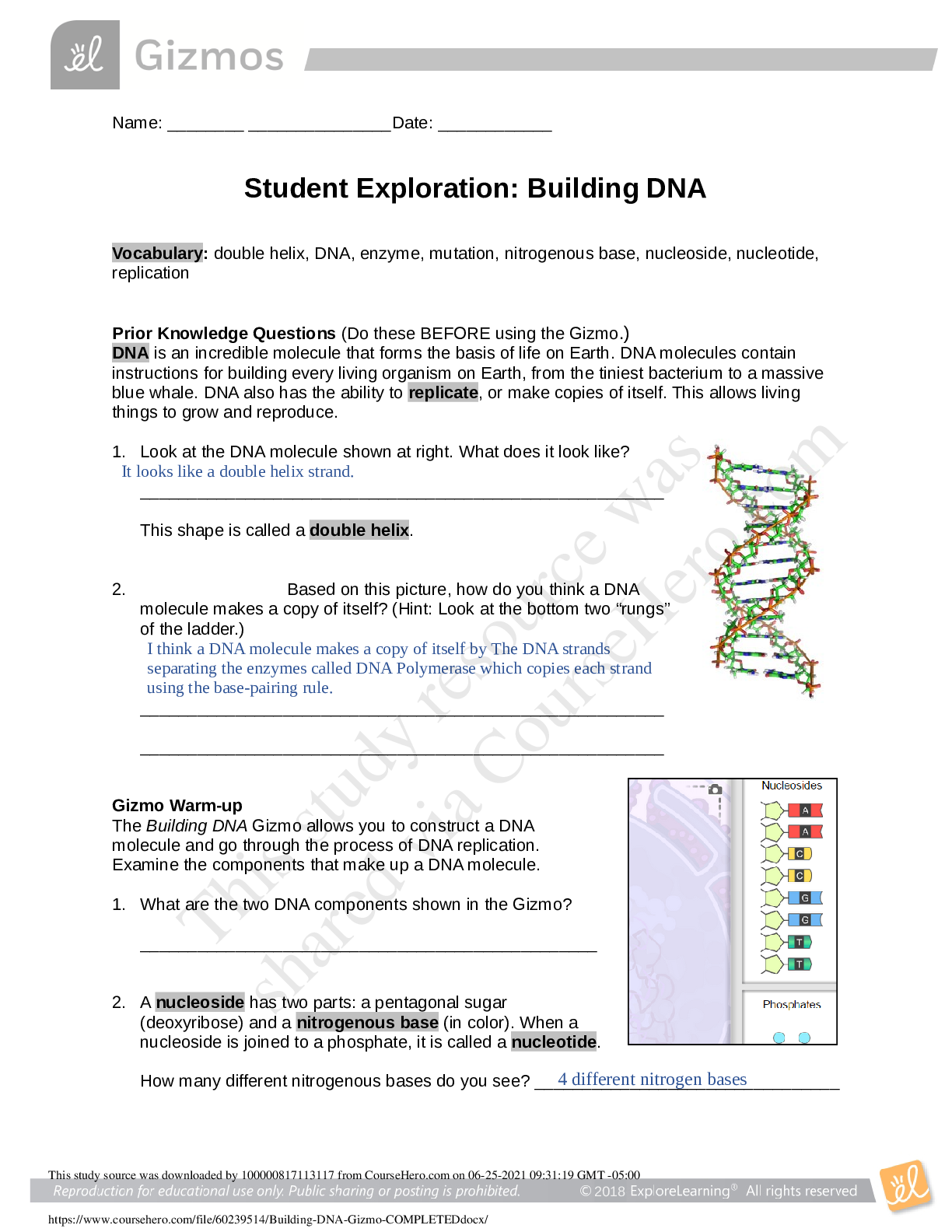
Document Content and Description Below
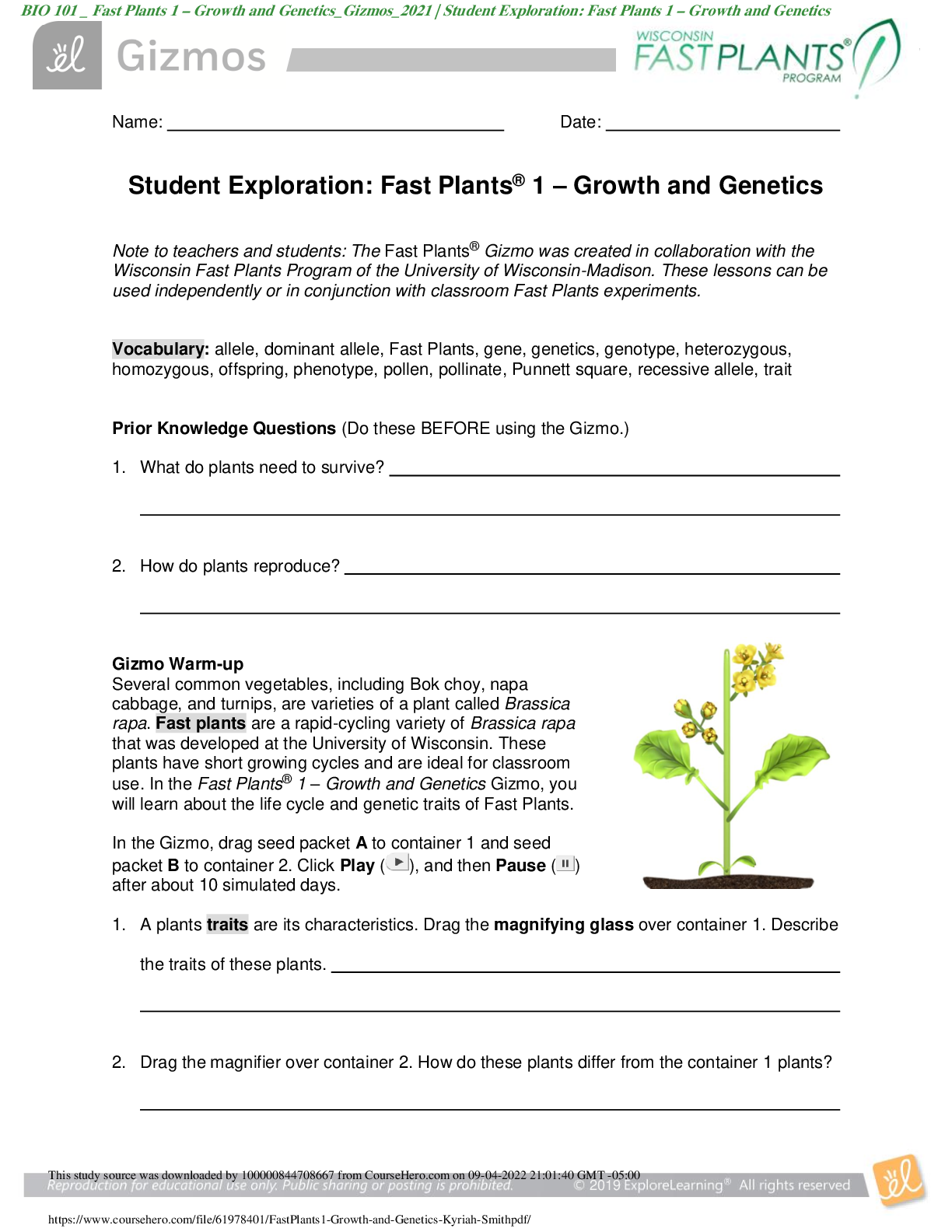
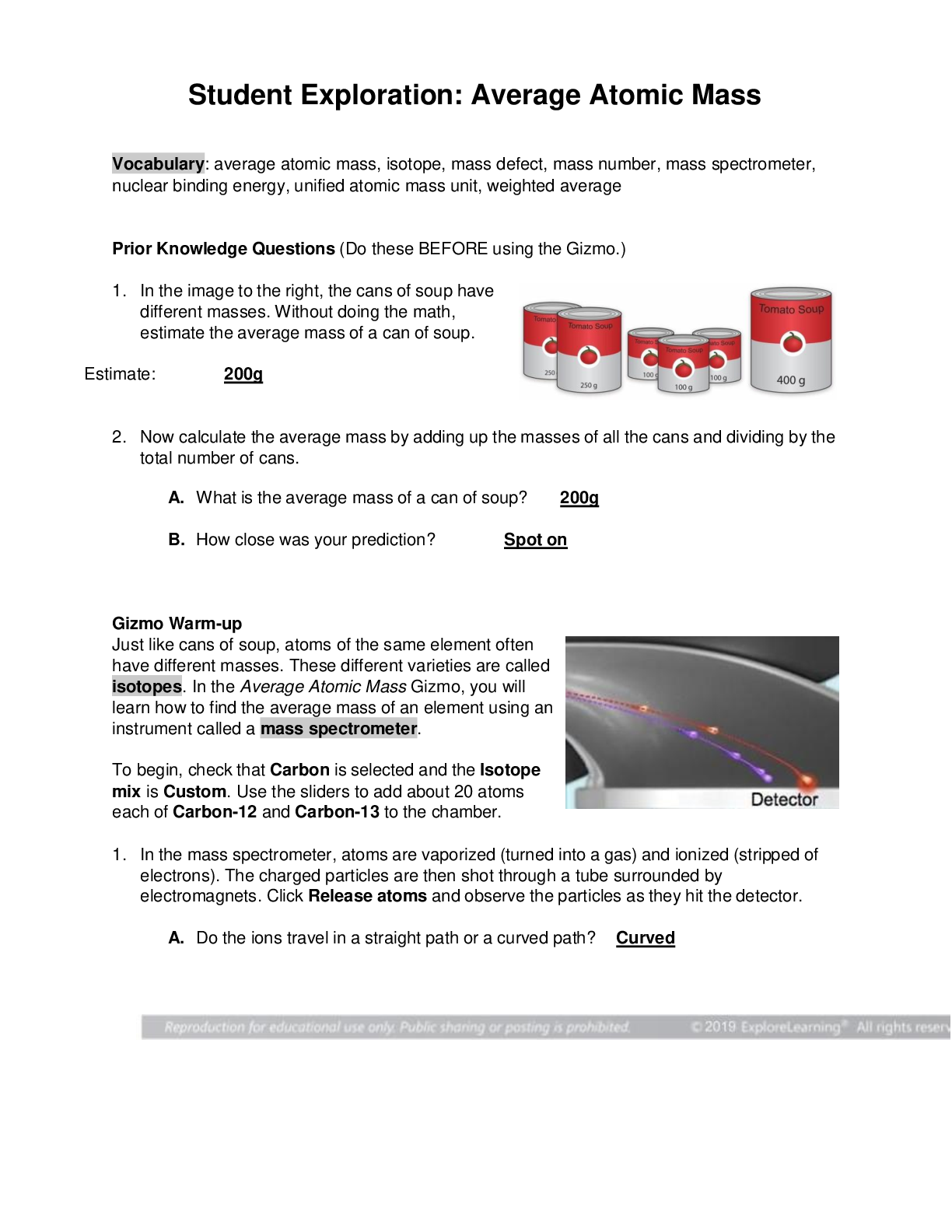
Vocabulary: average atomic mass, isotope, mass defect, mass number, mass spectrometer, nuclear binding energy, unified atomic mass unit, weighted average Prior Knowledge Questions (Do these BE... FORE using the Gizmo.) 1. In the image to the right, the cans of soup have different masses. Without doing the math, estimate the average mass of a can of soup. Estimate: 200g 2. Now calculate the average mass by adding up the masses of all the cans and dividing by the total number of cans. A. What is the average mass of a can of soup? 200g B. How close was your prediction? Spot on Gizmo Warm-up Just like cans of soup, atoms of the same element often have different masses. These different varieties are called isotopes. In the Average Atomic Mass Gizmo, you will learn how to find the average mass of an element using an instrument called a mass spectrometer. To begin, check that Carbon is selected and the Isotope mix is Custom. Use the sliders to add about 20 atoms each of Carbon-12 and Carbon-13 to the chamber. 1. In the mass spectrometer, atoms are vaporized (turned into a gas) and ionized (stripped of electrons). The charged particles are then shot through a tube surrounded by electromagnets. Click Release atoms and observe the particles as they hit the detector. A. Do the ions travel in a straight path or a curved path? Curved [Show More]
Last updated: 1 year ago
Preview 1 out of 9 pages
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Jun 26, 2021
Number of pages
9
Written in
Additional information
This document has been written for:
Uploaded
Jun 26, 2021
Downloads
0
Views
42



.png)
.png)
.png)


.png)
.png)
.png)















.png)