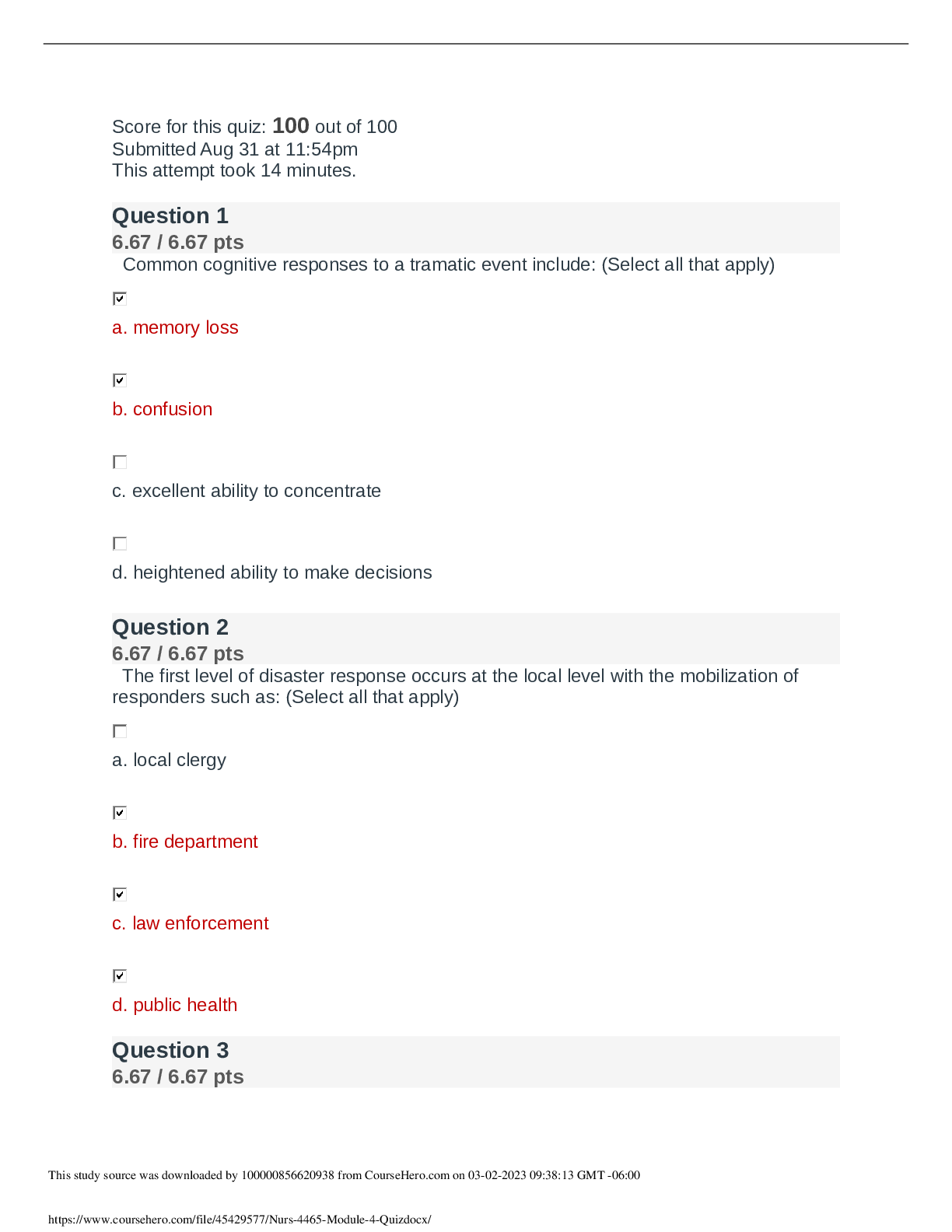
CHEM 120 Week 8 Final Exam Review (2022)
Document Content and Description Below
1. Question: Know what is the atomic number, atomic mass, protons, neutrons & electrons Atomic number – is the number of protons in the nucleus of an atom of any element Atomic mass – is expre... ssed in amu; is the wighted average Protons – are positively charged particles in the nucleus Neutron – are neutral, have no electrical charge Electrons – are negatively charged particles in the nucleus Atomic number = number of protons Atomic number – (minus) atomic mass = neutrons 2. Question: Describe the shapes of molecules H2S, CBr4, & PCl3 H2S – bent Number of bonded atoms: 2 Number of lone pairs: 2 Number of electron sets: 4 Number of bonded atoms: 4 Number of lone pairs: 0 Number of electron sets: 4 PCl3 – pyramidal Number of bonded atoms: 3 Number of lone pairs: 1 Number of electron sets: 4 3. Question: Calculating types of bonding (ionic, covalent, nonpolar covalent) Subtract the smaller EN value from the larger EN value 0 – 0.4 = nonpolar covalent > (greater than) 0.4 – 1.7 = polar covalent > (greater than) 1.7 = ionic bond Ionic bonds – is a bond that forms when electrons is being transferred from one form to another form, which results in positive & negative ions Covalent bond – a bond formed when atoms share electrons Nonpolar covalent bonds – when the EN is very similar between 2 atoms Polar covalent – when the EN is different between 2 atoms 4. Question: Non – polar covalent bonds & polar covalent bonds & electron sharing Bonding electron pair – the shared pair of electrons in covalent bonds Nonbonding pairs or lone pairs – the electrons that are not shared as part of the covalent bond Nonpolar covalent bonds – when the EN is very similar between 2 atoms Polar covalent – when the EN is different between 2 atoms 5. Question: Know the structure & makeup of DNA & RNA, know what DNA replication, transcription & translation are & ribosomes role in protein synthesis. Deoxyribonucleic acid (DNA) – provides a mechanism for heredity & serves as the blueprint for all the proteins of an organism The DNA helix is held together by hydrogen bonds between the nucleotides Adenine (A) binds with Thymine (T) in DNA Guanine (G) binds with Cytosine (C) in DNA & RNA DNA complimentary base pairs: A – T; C – G DNA is made up of genes that code for specific proteins Ribonucleic acid (RNA) – carries information needed to the protein synthesis Adenine (A) binds with Uracil (U) in RNA Guanine (G) binds with Cytosine (C) in DNA & RNA RNA complimentary base pairs: A – U; C – G DNA & RNA are polymers of repeating units of nucleotides DNA Replication – is copying or duplication of DNA molecules Transcription – the transfer of the DNA gene information to RNA which is transported out of the nucleus [Show More]
Last updated: 1 year ago
Preview 1 out of 7 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Reviews( 0 )
$10.00
Document information
Connected school, study & course
About the document
Uploaded On
Oct 18, 2022
Number of pages
7
Written in
Additional information
This document has been written for:
Uploaded
Oct 18, 2022
Downloads
0
Views
45



.png)



.png)

















