Test 2 (2021-2022). University of Notre Dame CHEMISTRY CHM2045
Document Content and Description Below
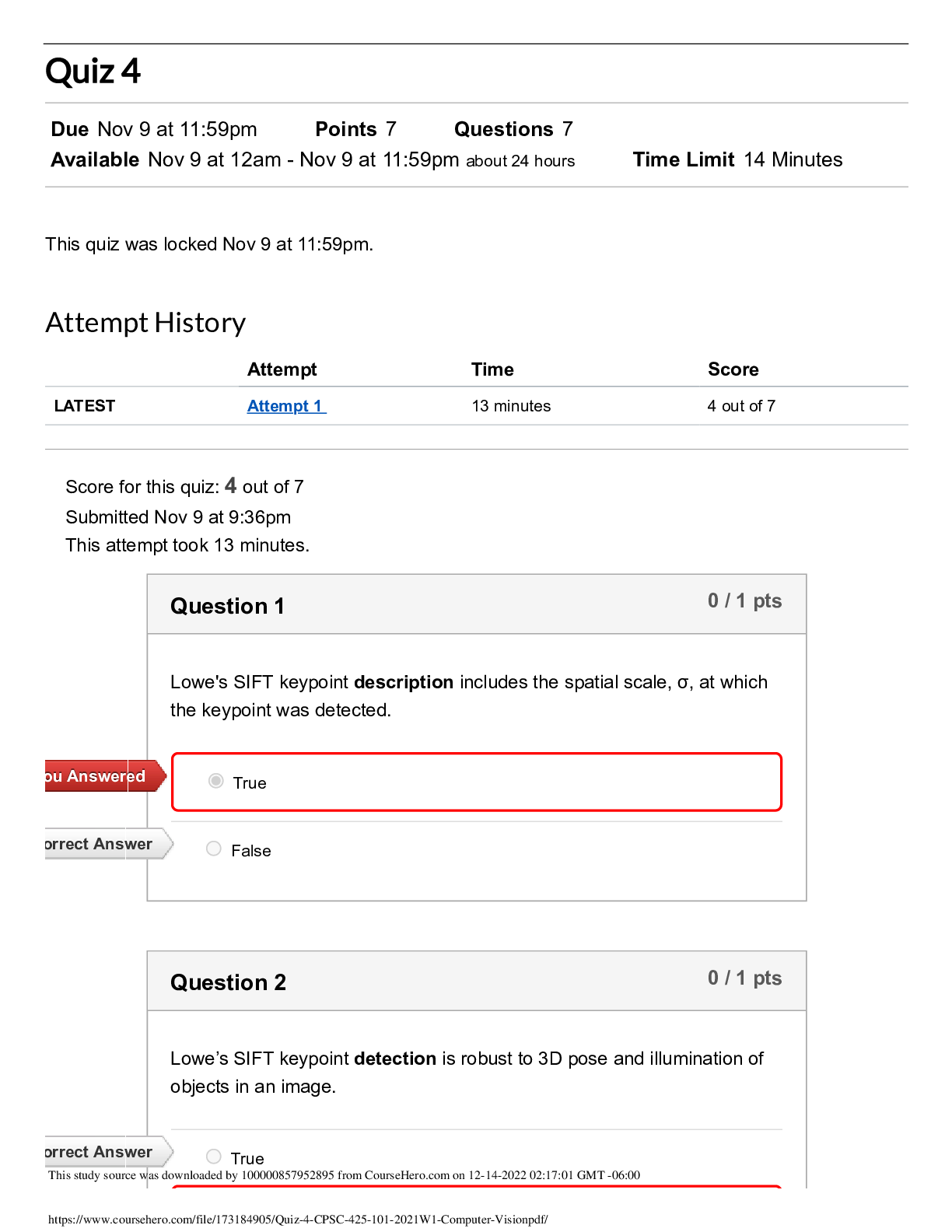
University of Guyana Lecturer: V. Mahabir Berbice Campus Date: 2nd December 2022 Department of Chemistry Duration: 2 hrs. CHM 2104 – Chemical Thermodynamics Test #2 Highlight in yellow or bold ... the correct answer for Section A. In Section B type responses for question 1 and write/type and/or snap photo of answer for question 2. Label/number all answers appropriately for Section B. Test is worth 28 marks which will correspond to 10% of the course. Section A 1. Solubility of a substance which dissolves with an increase in volume and liberation of heat will be favoured by the [1] (A)low pressure and high temperature. (C) low pressure and low temperature. (A)high pressure and low temperature. (D) high pressure and high temperature. 2. A chemical reaction will occur spontaneously at constant pressure and temperature, if the free energy is [1] (A)Zero (B) Positive (C) Negative (D) None of these 3. __________ increases with increase in pressure. [1] (A)The melting point of wax (C) The boiling point of a liquid (B) both (a) and (b) (D) neither (a) nor (b) 4. Degress of freedom at triple point will be [1] (A)0 (B) 1 (C) 2 (D) 3 5. When pressure is applied on the system, ice water, then [1] (A)equilibrium can not be established. (C) more ice will be formed. (B) more water will be formed. (D) evaporation of water will take place. 6. Partial molar free energy of an element A in solution is same as its [1] (A)chemical potential (B) activity (C) fugacity (D) activity coefficient 7. The temperature at which both liquid and gas phases are identical, is called the __________ point [Show More]
Last updated: 1 year ago
Preview 1 out of 3 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Reviews( 0 )
$4.00
Document information
Connected school, study & course
About the document
Uploaded On
Dec 14, 2022
Number of pages
3
Written in
Additional information
This document has been written for:
Uploaded
Dec 14, 2022
Downloads
0
Views
74



.png)


.png)
.png)


