CHEM 120 Week 4 Assignment: 20 points - Chamberlain College of Nursing
Document Content and Description Below
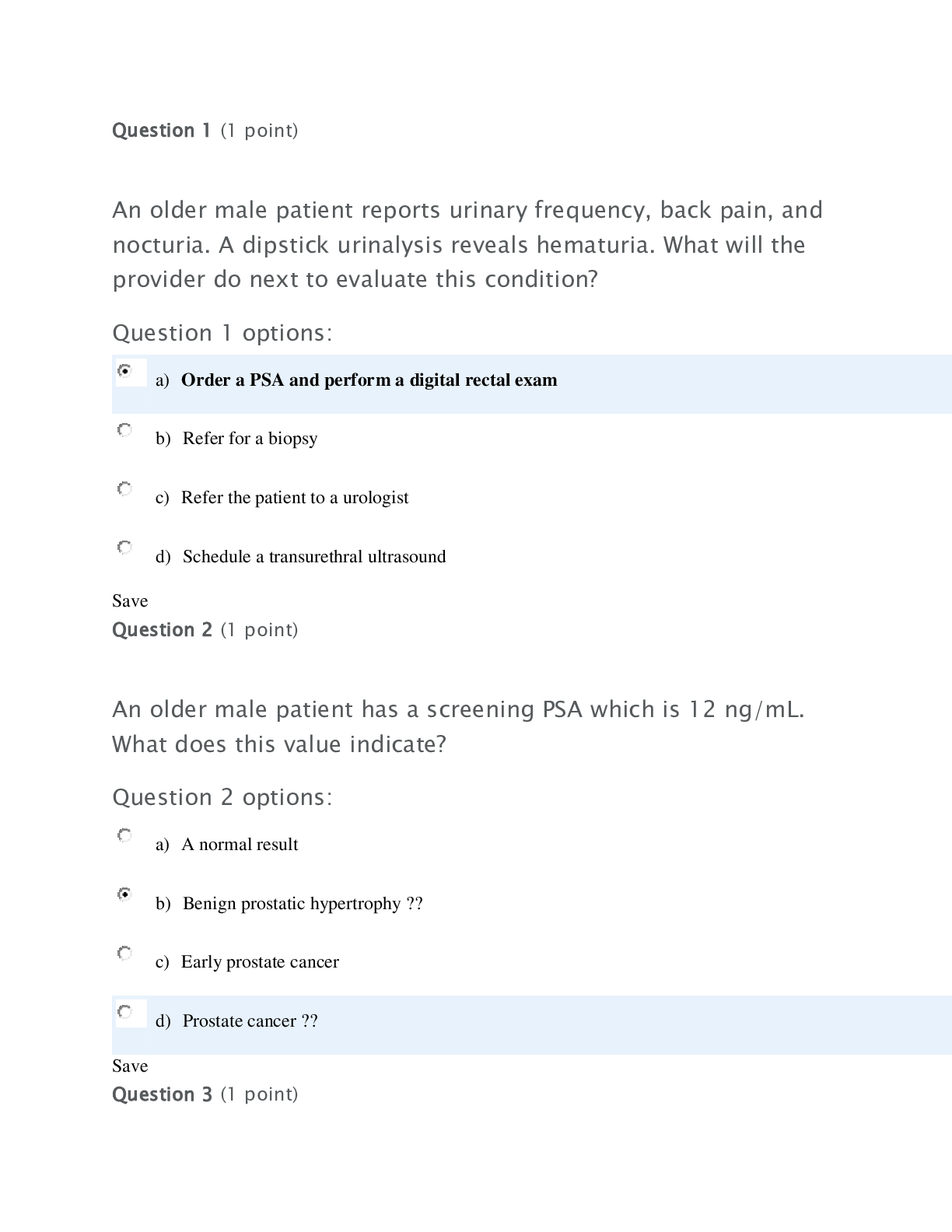
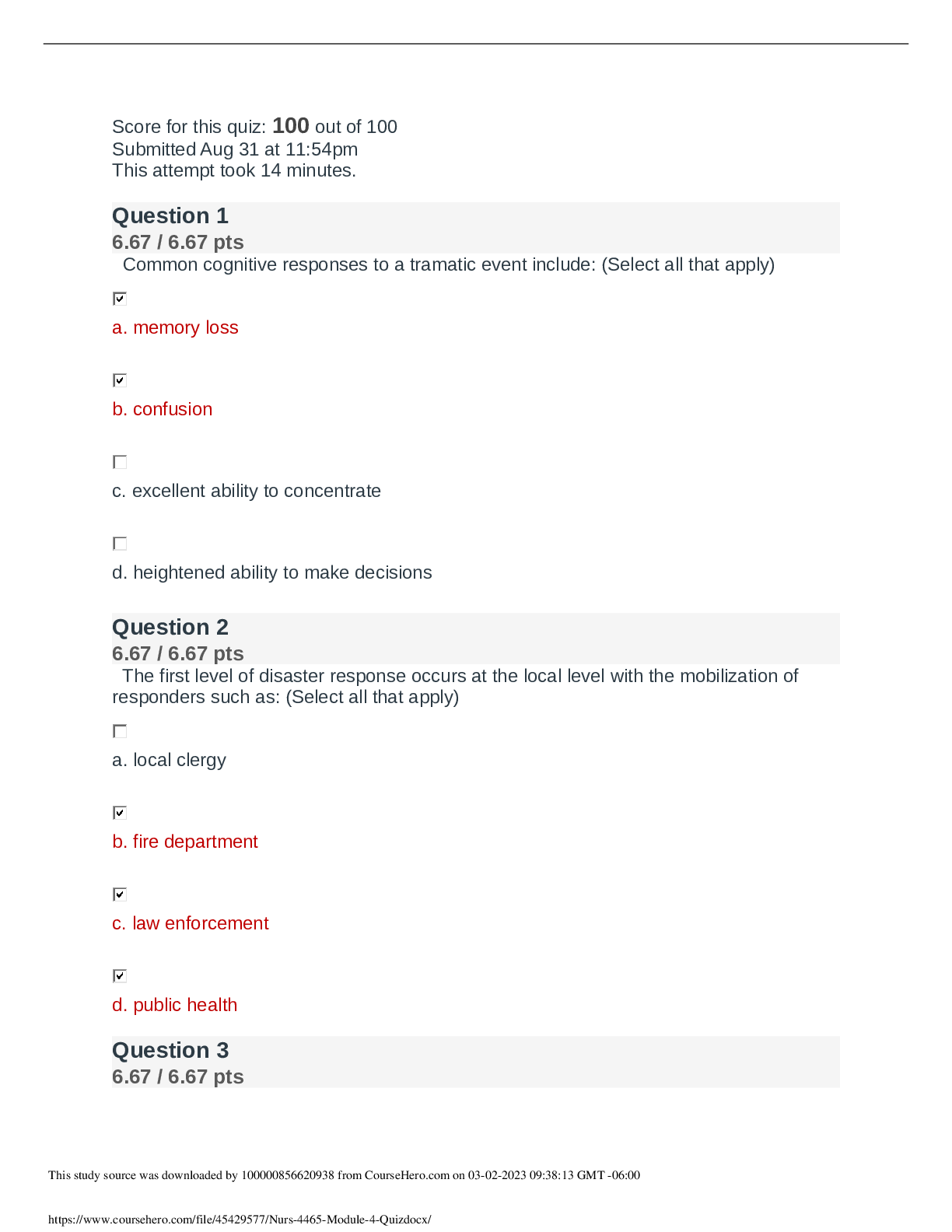
CHEM 120 Week 4 Assignment: 20 points.1. Solve these ideal gas law problems. Show all work for credit. a. A gas occupies 250 mL at 739 mmHg. For a gas at constant temperature, determine the volume ... of gas at 514 mmHg. (1 points) P*V=P*V 739*250 = 514*V 184750/514 = 359 b. Use the molar volume of a gas, at STP, to determine the number of moles of CO2 in 4.00 L of CO2 gas. (1 points) 22.4L/mol 4*(1/22.4) 0.179 c. A sample of nitrogen (N2) has a volume of 50.0 L at a pressure of 2 ATM. What is the volume of gas at a pressure of 15 atm, if there is no change in temperature? (1 points) 2*50 = 15*v V= 6.66 L d. A 0.6 L canister of Oxygen gas has a temperature of 297 K contains 21 mols of O2 gas. What is the pressure of this tank? (1 points) PV=nRT P*0.6=21*0.0821*297= P= 853 mmHg e. Calculate the pressure, in atmospheres, of 8.67 mol CO(g) in a 2.0 L tank at 48 degrees C. (1 points) PV=nRT 48+273.15= 321.15 K P*2=8.67*0.0821*321.15 P= 114 mmHg 2. According to the Bronstead Lowery acid/base theory, what is the definition of an acid and a base? (2 points) [Show More]
Last updated: 1 year ago
Preview 1 out of 3 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Reviews( 0 )
$8.00
Document information
Connected school, study & course
About the document
Uploaded On
Nov 22, 2021
Number of pages
3
Written in
Additional information
This document has been written for:
Uploaded
Nov 22, 2021
Downloads
0
Views
90



.png)



.png)