BioChemistry > QUESTIONS & ANSWERS > BCHM 307 Exam test questions and answers solution docs 2020 (All)
BCHM 307 Exam test questions and answers solution docs 2020
Document Content and Description Below
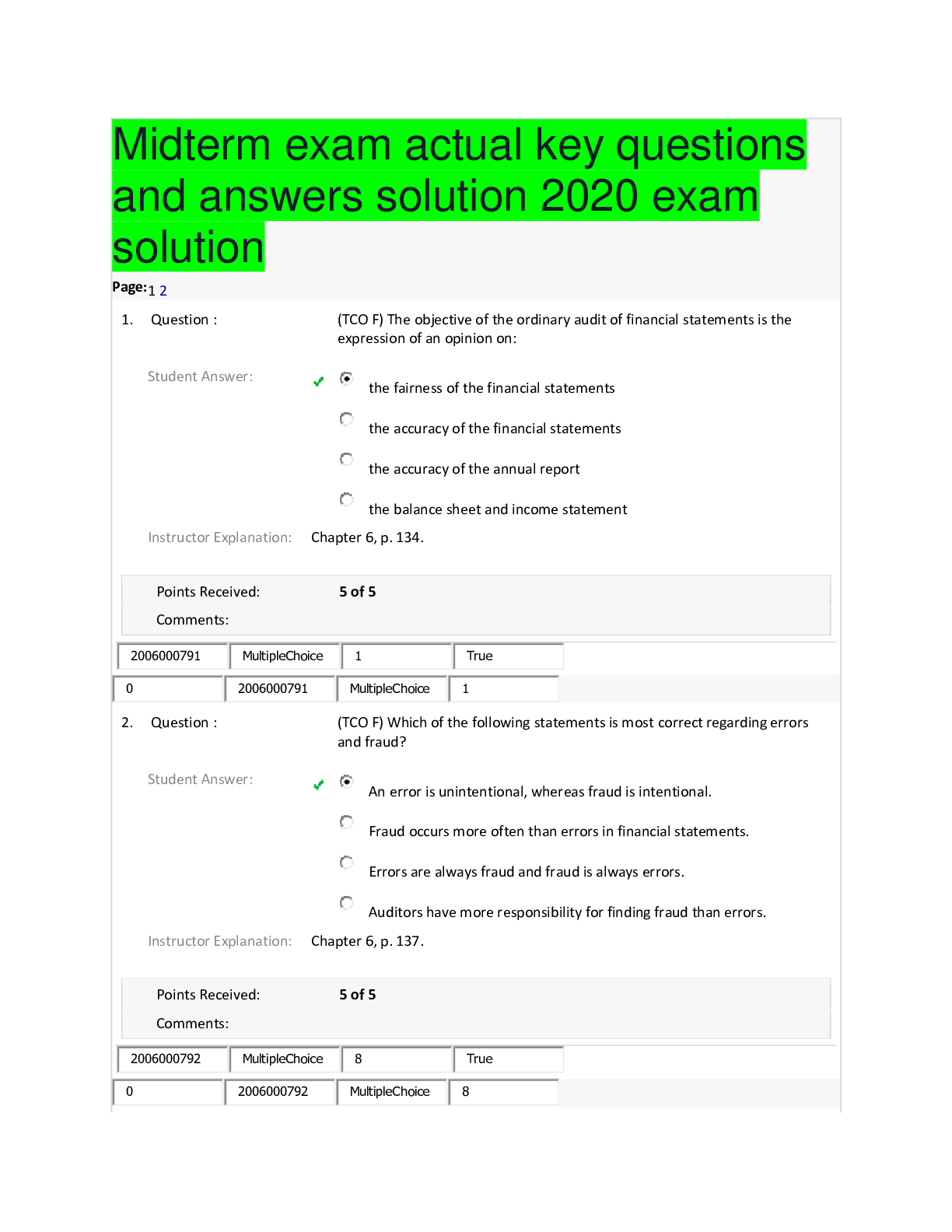
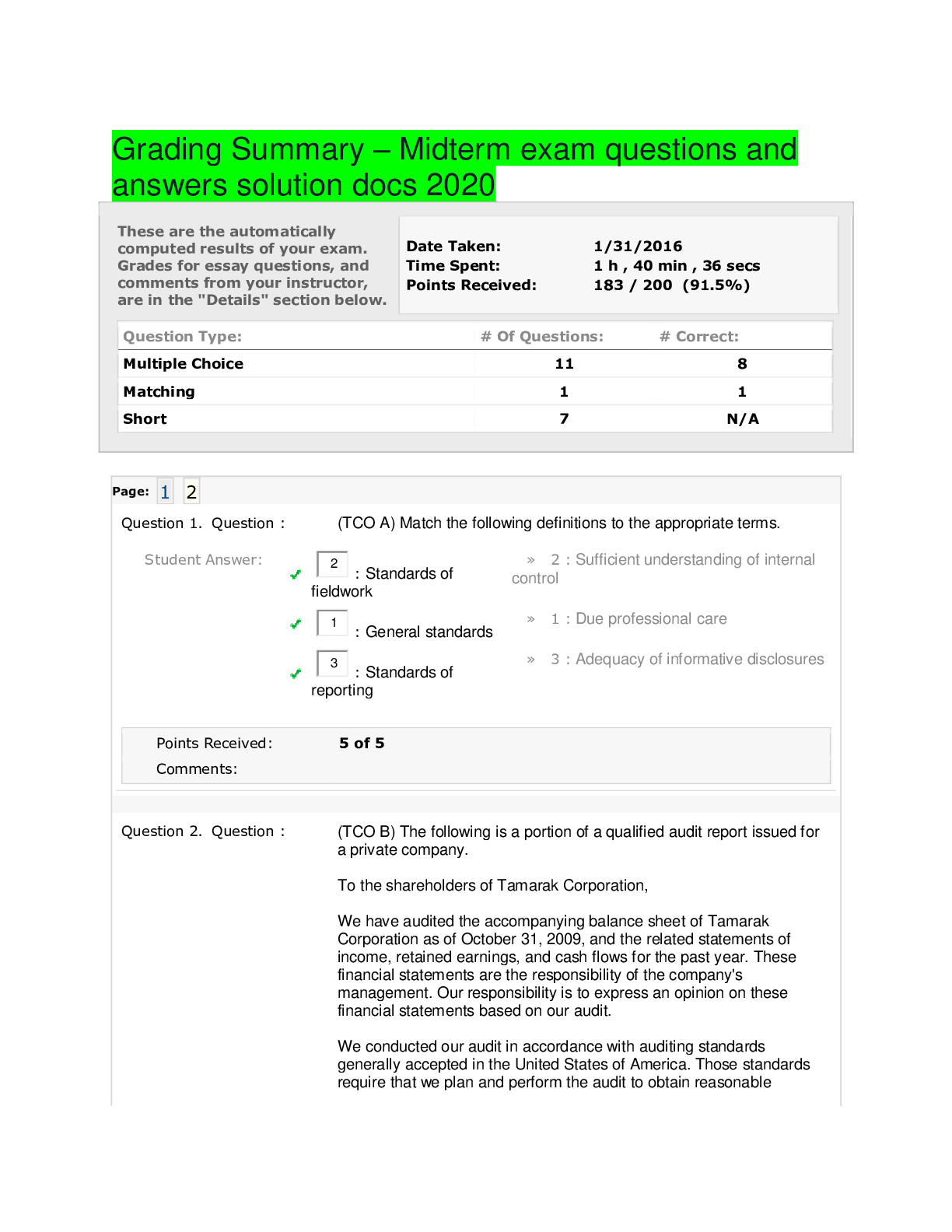
BCHM 307 Exam test questions and answers solution docs 2020 Which of the major types of biomolecules is never found in a polymeric form? An exergonic process Which of the following is a major diff... erence between eukaryotic and prokaryotic cells? Which elements are found in simple carbohydrates? ΔG° and ΔG mean the same thing. If a reaction at 37°C has a ΔH of 23 kJ/mol and a ΔS of 337 J/K•mol, what is the ΔG for the reaction? Which of the following biopolymers is correctly paired with the bond that forms between the monomers? Simple molecules condense to form more complex molecules called: Which of the following is the most abundant element in the human body? A spontaneous process always has Entropy is used to measure A ΔGo value of zero indicates that the reactions or processes are at equilibrium. Which of the following molecules contains the most oxidized form of carbon? Which of the biopolymers is correctly paired with its major function? A gaseous mixture of hydrogen, water, ammonia and methane can produce which of the biomolecules when exposed to an electrical discharge (such as lightening)? Photosynthetic organisms use energy from the sun to reduce _____ to _____. Which of the following explains how nucleotides might have polymerized into nucleic acids in the prebiotic world? Most living matter consists of a relatively large number of chemical elements. If the following two reactions were coupled, what would be the ΔG for the overall exergonic reaction? ATP + H2O -------> ADP + Pi [ ΔG = -31 kJ/mol ] Glucose + Pi --------> glucose-1-phosphate + H2O [ ΔG = 21 kJ/mol ] The similarity of one organism to another (for example a bacteria versus a human) is most easily done by comparing which biopolymer? Quiz 2 Which of the following is an example of an amphipathic molecule? If a phosphate buffer (pK=6.82) was formulated such that its pH was 7.3, it would be best suited to buffer against _____. If instead, it was formulated such that its pH was 6.3, it would be best suited to buffer against _____. The strongest non-covalent interactions are _____. Which of the following explains the interactions that occur between the atoms of water molecules and the ions that form when sodium chloride dissolves in water? Which of the following would be the strongest acid? In a hydrogen bond between a water molecule and another biomolecule, _____. Which of the following is true regarding hydrophobic interactions between nonpolar molecules or groups? Which of the following molecules would be prevented from readily crossing a lipid bilayer? Which of the following explains the attractive forces between hydrophobic molecules in an aqueous solution? A molecule that has both a polar and nonpolar region is called _____________. Due to the formation of hydrogen bonds, _____ is highly soluble in water. Which of the following is a physical property of water that results from hydrogen bonding? Which of the following functional groups has two hydrogen bond donors and one hydrogen bond acceptor? In a water molecule, hydrogens are partially _____; oxygens are partially _____. What term is used to describe the exclusion of nonpolar substances from an aqueous solution? When a non-polar substance is added to water, how do the molecules of water behave? At any given moment, data suggests that a single (liquid) water molecule participates in _____ strong hydrogen bond(s) at any given time. The role played by the water molecule is best characterized as _____. Metabolic acidosis often causes increased respiratory rates. What portion of the bloodstream buffer is lost through increased respiration? Which of the following is an example of the hydrophobic effect? The results of DNA sequencing are obtained by first separating different sized pieces of DNA using ________ followed by detection of the particular dideoxynucleotide using _______. The purines found in nucleic acids are __________ and ___________. The coenzymes known as NAD+, FAD and coenzyme A all contain a derivative of ________. The presence of ________ approximately once in every 1000 base pairs is what makes each individual human genetically unique. The Because of the high temperature ______ step during a PCR reaction, the DNA polymerase from ____ is used. What term is used to describe short segments of DNA that are copied many times and inserted randomly in chromosomes. What reagents are required to perform PCR? If instead of four different bases in DNA and RNA, there were six, what is the minimum size of a codon to encode the 20 amino acids commonly found in proteins? The triplet code allows many amino acids to be specified by more than one codon. Such a code is said to be _________. What term describes the process of converting the information found in DNA into the sequence of a protein Which of the following non-covalent interactions is the most important in maintaining the structure of the double helix? The 5' end of a polynucleotide contains _____, while the 3' end contains ________. Which of the following is a characteristic of Chargaff's rule? A genomic library __________. [Show More]
Last updated: 1 year ago
Preview 1 out of 6 pages
Instant download

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Nov 01, 2020
Number of pages
6
Written in
Additional information
This document has been written for:
Uploaded
Nov 01, 2020
Downloads
0
Views
93



















.png)
 ans.png)
.png)


