Chemistry > Experiment > Exploring the behavior of Gases (All)
Exploring the behavior of Gases
Document Content and Description Below
Part I 1. Go to the intro. Choose only one type of particle. 2. Record the pressure, temperature and volume. 3. Give one pump of gas and observe the behavior. How would you describe this? Record t... he pressure, temperature and volume. 4. Hold volume and temperature constant and give one more pump. Record the number of particles and the pressure in the data table below. Describe what you saw. Repeat this a few times, either increasing or decreasing the number of the same type of particles. Number of particles Pressure (atm) 5. Is there a relationship between the number of particles and the pressure? Briefly describe this. What is the pressure in the container due to? (what assumption are we making?) Modified resource from https://phet.colorado.edu/en/simulation/gases-intro 2 Part II: There are 3 parameters that need to be specified when describing a specific quantity of a gas. They are: Pressure, Volume and Temperature. We will keep the number of particles constant in each “experiment” and explore the effect (if any) a change in any of these parameters may have on the behavior of the gas. Choose the Laws option on the right. See picture Experiment 1- Volume 1. Give one pump of gas into the chamber. 2. Choose to hold the volume constant by selecting that option in the upper right-hand corner. See the picture. What is the initial temperature (in K) and pressure (in atm) in the chamber? 3. Use the slider at the bottom of the simulator to add heat and double the temperature. Did the pressure go up or go down? What is the new pressure in the chamber? 4. Keeping the volume constant (and the number of particles constant), change the temperature, and record the pressure. Repeat 4 times and record your data. Sketch a graph to the right of the table. Be sure to give a title to your graph and label the axis completely. Independent variable is : _________________ Dependent Variable: ____________________ Constants: ____________________________ Temperature (K) Pressure (atm) Describe the graph and relationship: This study source was downloaded by 100000830919685 from CourseHero.com on 06-06-2022 16:28:36 GMT -05:00 https://www.coursehero.com/file/59856218/Exploring-the-behavior-of-Gases-Phetpdf/Honors Chemistry Srikanthan Modified resource from https://phet.colorado.edu/en/simulation/gases-intro 3 Experiment 2 - Temperature 1. Reset the simulator by selecting the reset button in the bottom right corner of the simulation. 2. Give one pump of gas into the chamber. 3. Choose to hold the temperature constant by selecting that option in the upper right-hand corner. See the picture. What is the initial pressure (in atm) in the chamber? [Show More]
Last updated: 1 year ago
Preview 1 out of 5 pages

Reviews( 0 )
Recommended For You
Biology> Experiment > Comparing DNA Sequences to Understand Evolutionary Relationships with Blast Purpose: The reason for doing this lab is to create cladograms that depict evolutionary relationships and to analyze biological data with an online bioinformatics tool. (All)

Comparing DNA Sequences to Understand Evolutionary Relationships with Blast Purpose: The reason for doing this lab is to create cladograms that depict evolutionary relationships and to analyze biological data with an online bioinformatics tool.
AP Biology Comparing DNA Sequences to Understand Evolutionary Relationships with Blast Purpose: The reason for doing this lab is to create cladograms that depict evolutionary relationships and to...
By QuizMaster , Uploaded: Mar 02, 2021
$9.5
Engineering> Experiment > Deposition of dielectric thin films RF Magnetron Sputtering MM 731: Experiments in advanced materials processing (Lab.) (All)

Deposition of dielectric thin films RF Magnetron Sputtering MM 731: Experiments in advanced materials processing (Lab.)
Deposition of dielectric thin films RF Magnetron Sputtering MM 731: Experiments in advanced materials processing (Lab.)RF (radio frequency) sputter deposition • Good for insulating materials beca...
By A-LEVEL GURU , Uploaded: Aug 25, 2022
$10
Chemistry> Experiment > PE 317 Unit operations Liquid –liquid extraction (All)
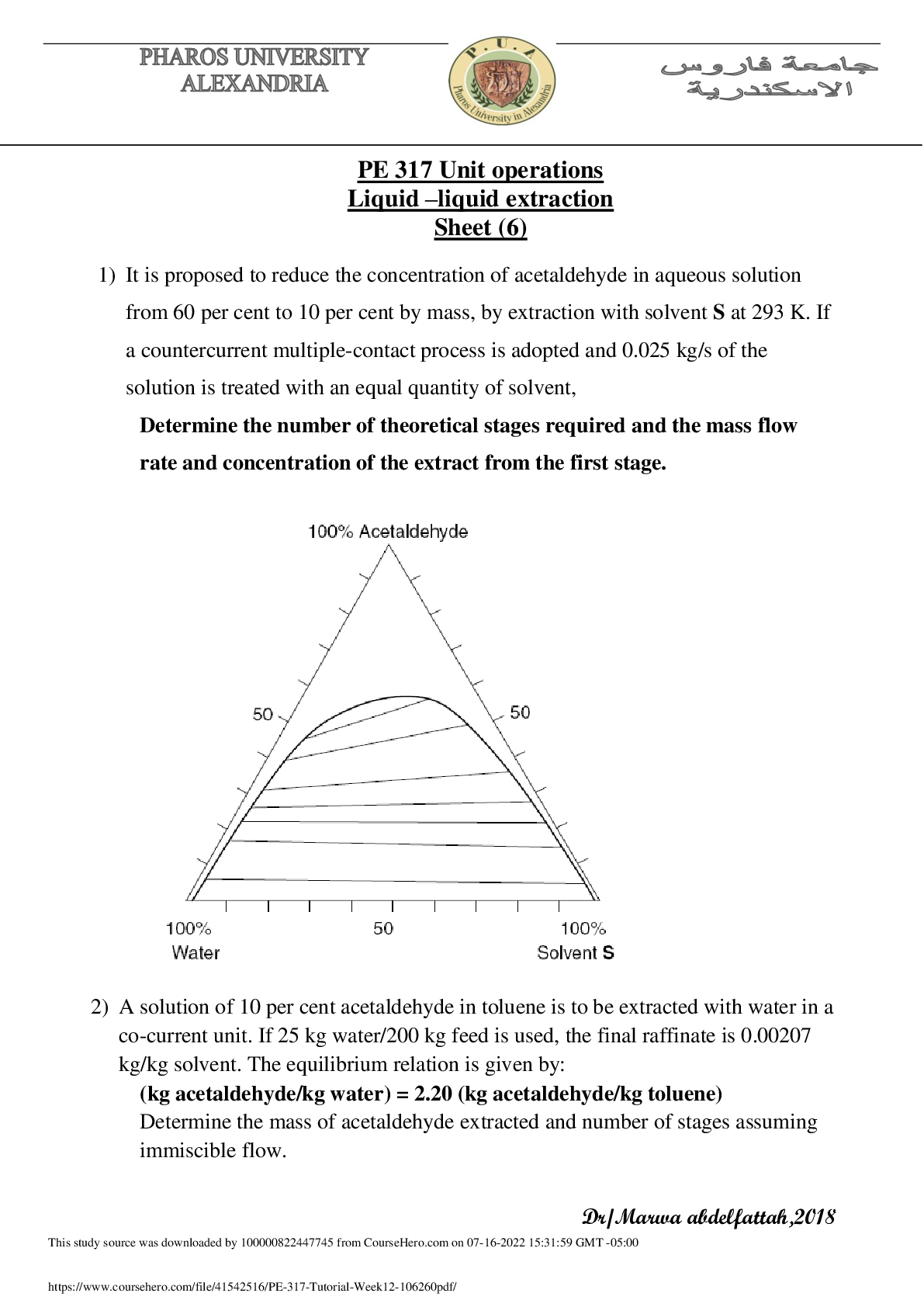
PE 317 Unit operations Liquid –liquid extraction
It is proposed to reduce the concentration of acetaldehyde in an aqueous solution from 60 percent to 10 percent by mass, by extraction with solvent S at 293 K. If a countercurrent multiple-contact p...
By CourseWorks,Inc , Uploaded: Jul 26, 2022
$7.5
Electrical Engineering> Experiment > Lab 1: Introduction to Electrical Measurements Oren Shed Matthew Amezaga ECEN 214 – 522 TA: Meltem Apaydin Lab Date: January 31, 2020 Due Date: February 7, 2020 (All)

Lab 1: Introduction to Electrical Measurements Oren Shed Matthew Amezaga ECEN 214 – 522 TA: Meltem Apaydin Lab Date: January 31, 2020 Due Date: February 7, 2020
Lab 1: Introduction to Electrical Measurements Oren Shed Matthew Amezaga ECEN 214 – 522 TA: Meltem Apaydin Lab Date: January 31, 2020 Due Date: February 7, 2020Procedure(theory) Task 1 of the...
By AMAZING GRACE , Uploaded: Jun 18, 2022
$6
Education> Experiment > Copy of Project 4_ Sound Waves - Lab Report (All)

Copy of Project 4_ Sound Waves - Lab Report
Copy of Project 4_ Sound Waves - Lab Report
By Nancylect , Uploaded: May 26, 2022
$7
Programming> Experiment > ISM 4210 ISM4210_LESSON7 DSC. (All)

ISM 4210 ISM4210_LESSON7 DSC.
Consider a real-life business situation and discuss under what circumstances you would consider using the MERGE statement. In addition to detail explanation, provide an SQL example to merge at least t...
By SuperSolutions© , Uploaded: Oct 13, 2020
$6.5
Physics> Experiment > Texas A&M University - ENGR 217 Lab 02; Electric Potential & Electric Fields (All)

Texas A&M University - ENGR 217 Lab 02; Electric Potential & Electric Fields
Electric Potential & Electric Fields. Abstract This experiment was performed with the purpose of finding and plotting the electric field vector components across a two-dimensional surface. The e...
By QuizMaster , Uploaded: Sep 16, 2020
$8.5
Statistics> Experiment > Statistics, 4th edition By David Freedman, Robert Pisani. (All)

Statistics, 4th edition By David Freedman, Robert Pisani.
Statistics, 4th edition By David Freedman, Robert Pisani.
By Grade A+ , Uploaded: Feb 18, 2022
$13
Chemistry> Experiment > CHEM 162/ CHEM 162 Experiment #5: Spectrochemical Series complete solution (All)

CHEM 162/ CHEM 162 Experiment #5: Spectrochemical Series complete solution
CHEM 162/ CHEM 162 Experiment #5: Spectrochemical Series complete solutionCHEM 162/ CHEM 162 Experiment #5: Spectrochemical Series complete solutionCHEM 162/ CHEM 162 Experiment #5: Spectrochemical...
By VERIFIED A+ , Uploaded: Dec 04, 2021
$9
Biology> Experiment > BIOD 171 M6: Module 6 Exam- Requires Respondus LockDown Browser: Essential Microbiology with Lab- Straley | Portage Learning (All)

BIOD 171 M6: Module 6 Exam- Requires Respondus LockDown Browser: Essential Microbiology with Lab- Straley | Portage Learning
M6: Module 6 Exam- Requires Respondus LockDown Browser Due No due date Points 100 Ques!ons 30 Time Limit 60 Minutes Requires Respondus LockDown Browser This quiz is currently locked. A!empt Histor...
By PHISHER , Uploaded: Oct 18, 2021
$11
Document information
Connected school, study & course
About the document
Uploaded On
Aug 06, 2022
Number of pages
5
Written in
Additional information
This document has been written for:
Uploaded
Aug 06, 2022
Downloads
0
Views
119






