BioChemistry > EXAM > BI/CH421 Biochemistry Exam 1 10/02/2017 | SOLVED EXAM, DOWNLOAD FOR QUALITY GRADES (All)
BI/CH421 Biochemistry Exam 1 10/02/2017 | SOLVED EXAM, DOWNLOAD FOR QUALITY GRADES
Document Content and Description Below
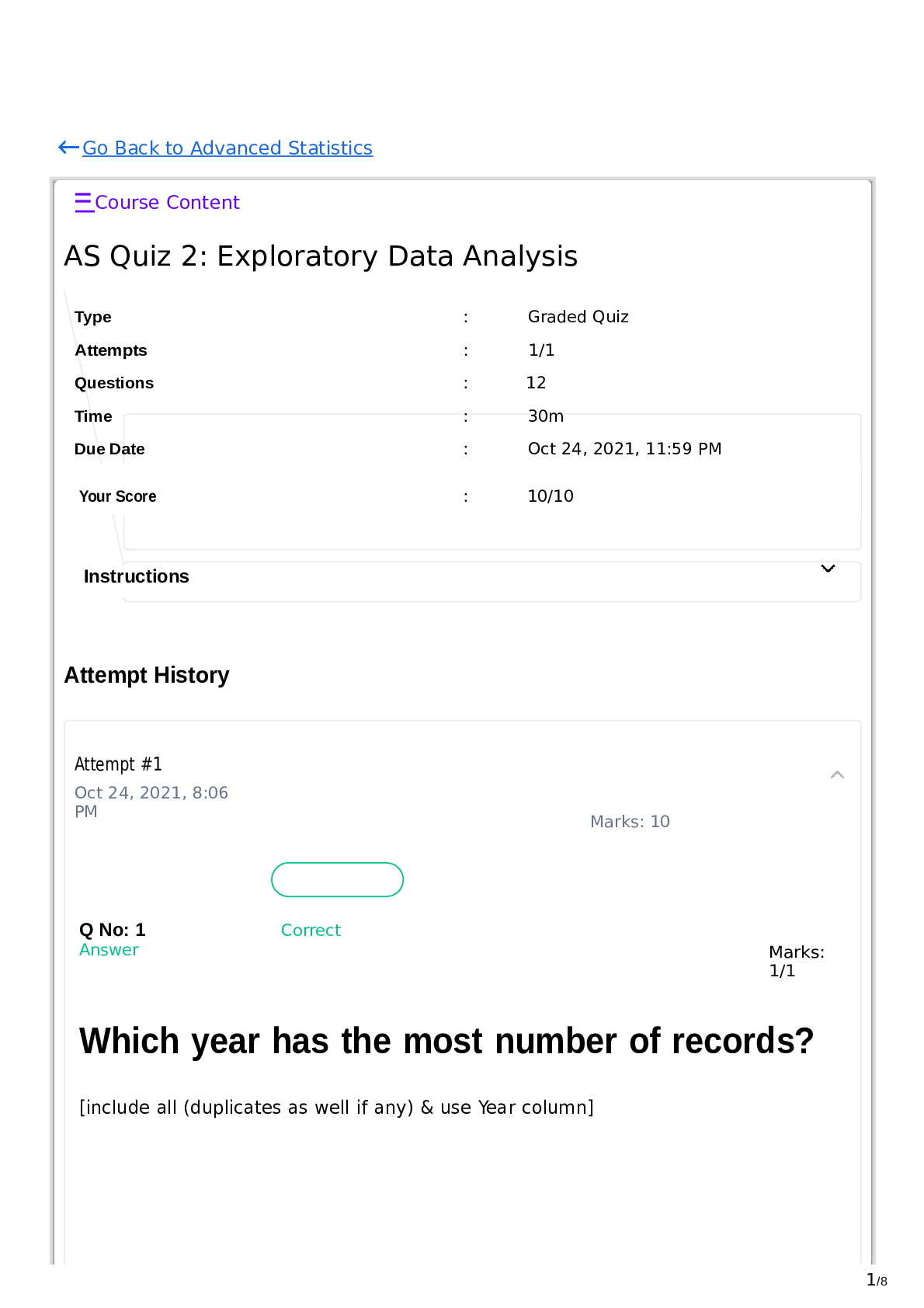
1. The equilibrium constant, Keq, for the hydrolysis of ATP (ATP à ADP + Pi) is 2 x 105 M. If the measured concentration of the cellular concentrations of ATP, ADP, and Pi are 5 mM, 0.5 mM and 5 ... mM respectively. From this information, you can say (worked example 1.1in your text) a. The reaction is at equilibrium inside cells b. The hydrolysis reaction will proceed spontaneously inside cells c. The hydrolysis of ATP will require an input of energy to occur within cells d. The hydrolysis of ATP could only occur spontaneously if it is coupled to an energetically favorable reaction e. Both C and D are true 2. Rank the following bonds in terms of their relative typical strengths 1: Dipole-dipole interaction 2: Ionic interaction 3: London dispersion forces 4: Hydrogen bond a. 4 > 1 > 2 > 3 b. 3 > 2 > 1 >4 c. 2 > 4 > 3 > 1 d. 2> 4 > 1 > 3 3. Which amino acid pairs listed below are some of the major contributors to protein absorbance at 280 nm? a. Tyrosine and proline b. Tryptophan and histidine c. Histidine and proline d. Tyrosine and tryptophan e. All amino acids absorb at 280 nm 4. How many hydrogen bonds can pyridoxal phosphate (shown below) donate to the surrounding water? a. 0 b. 1 c. 2 d. 3 e. >3 5. The pH of a solution containing 1x10-4 M of HCl will b [Show More]
Last updated: 1 year ago
Preview 1 out of 14 pages

Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
We Accept:

Also available in bundle (1)

BIOCHEMISTRY SOLVED EXAMS
Download a bundle of Biochemistry solved examination past papers at a quality price for quality grades. Thank you.
By Wanjiku 1 year ago
$45
5
Reviews( 0 )
$12.00
Document information
Connected school, study & course
About the document
Uploaded On
Oct 28, 2022
Number of pages
14
Written in
Additional information
This document has been written for:
Uploaded
Oct 28, 2022
Downloads
0
Views
46