Programming > EXAM > SOLVED - Elaborated SOCRA Certification Exam Final Graded A+ for 2023 (All)
SOLVED - Elaborated SOCRA Certification Exam Final Graded A+ for 2023
Document Content and Description Below
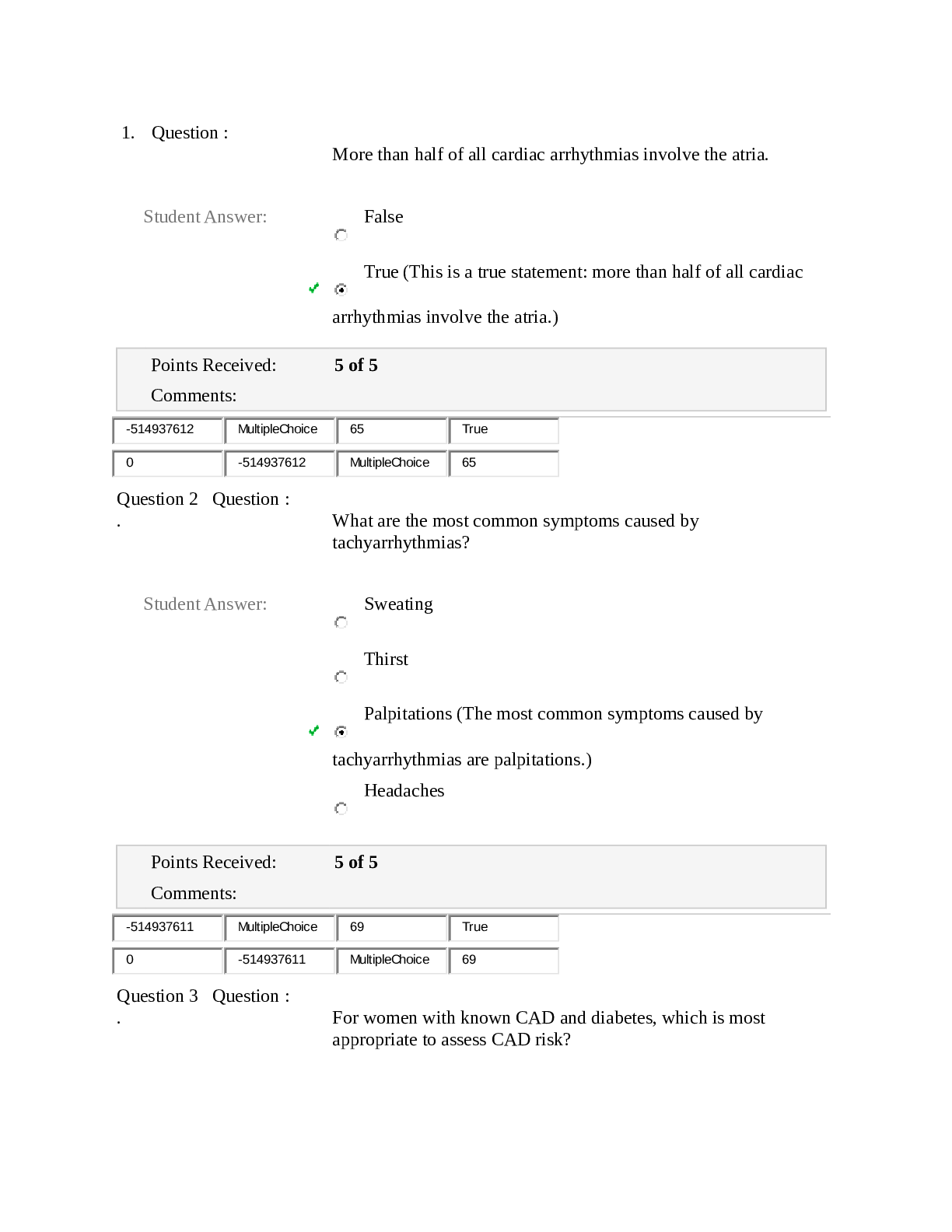
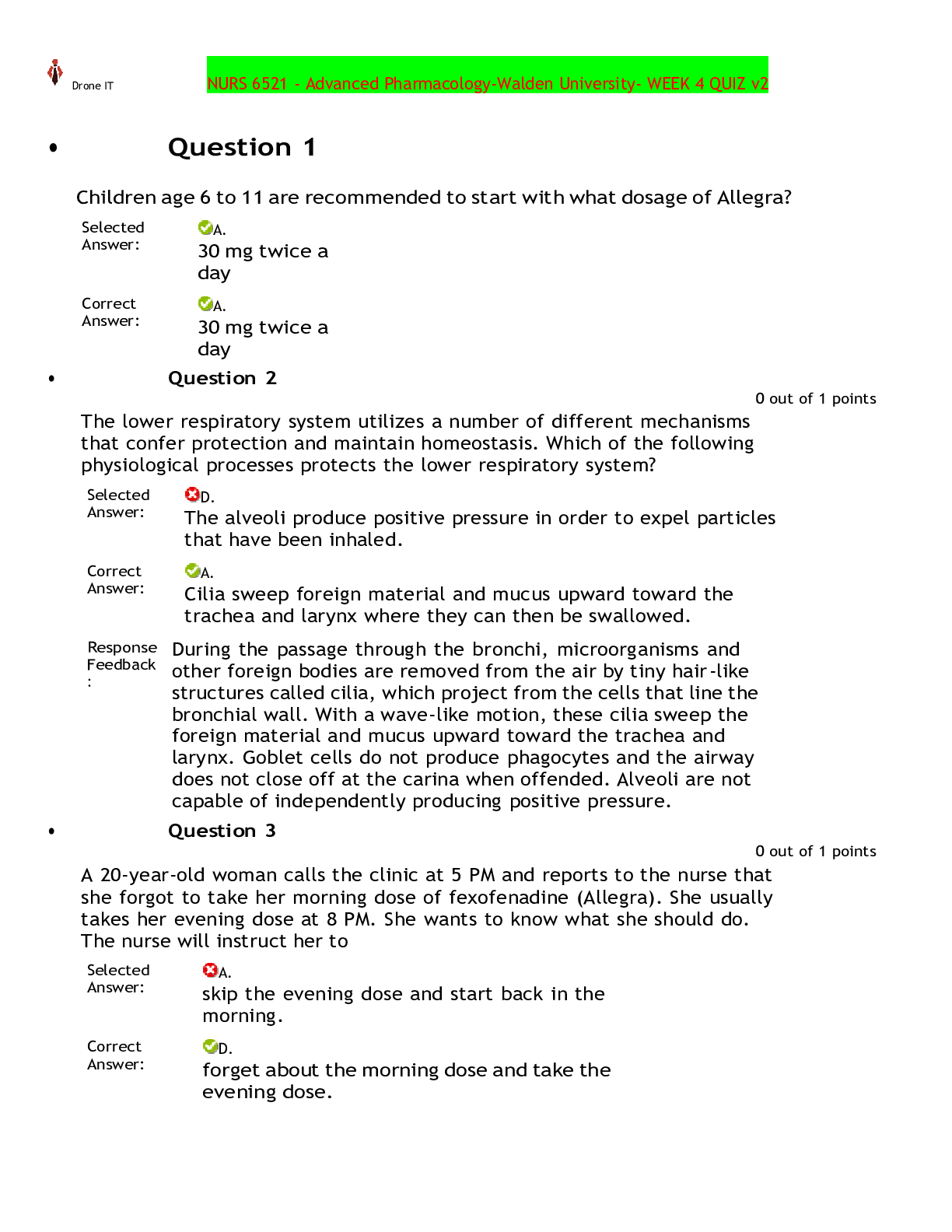
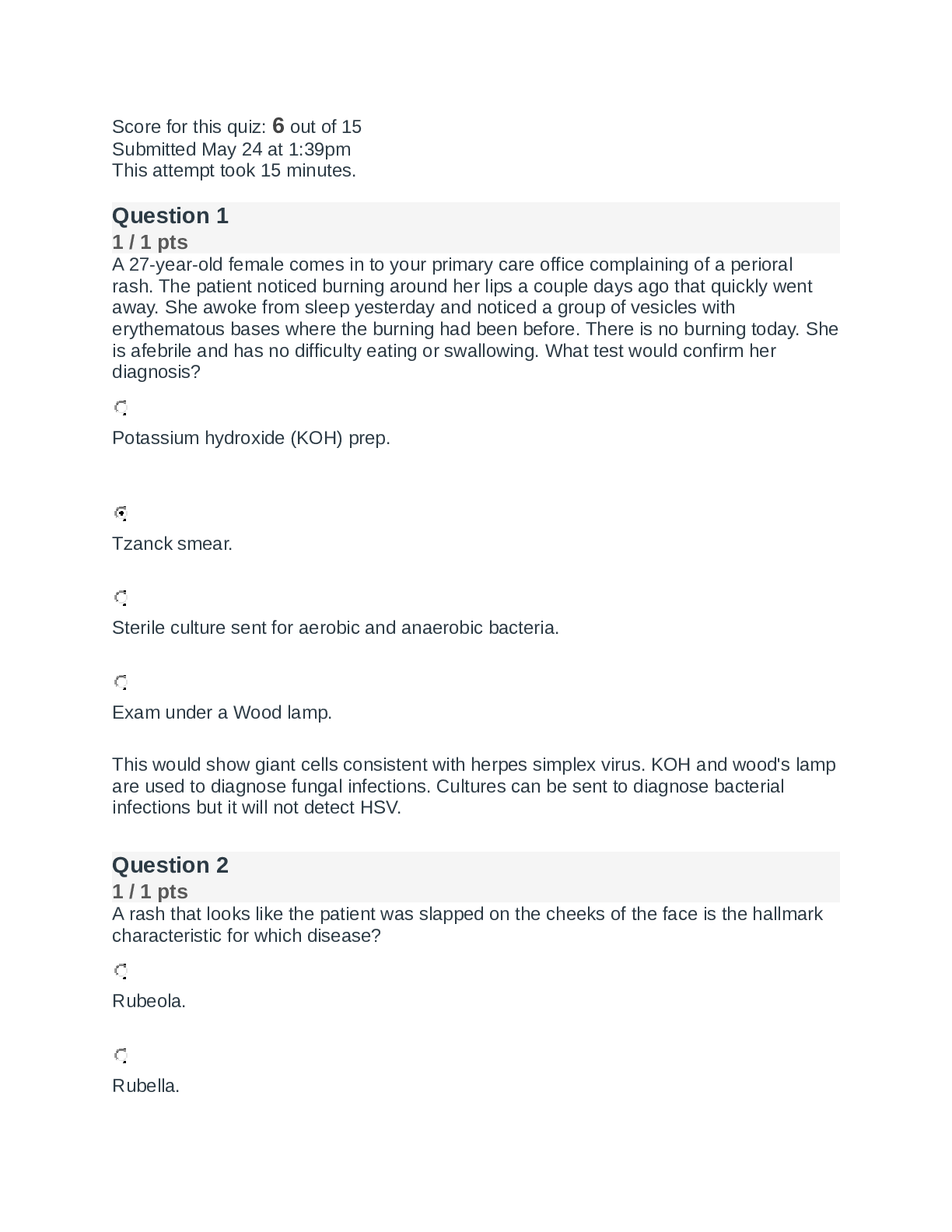
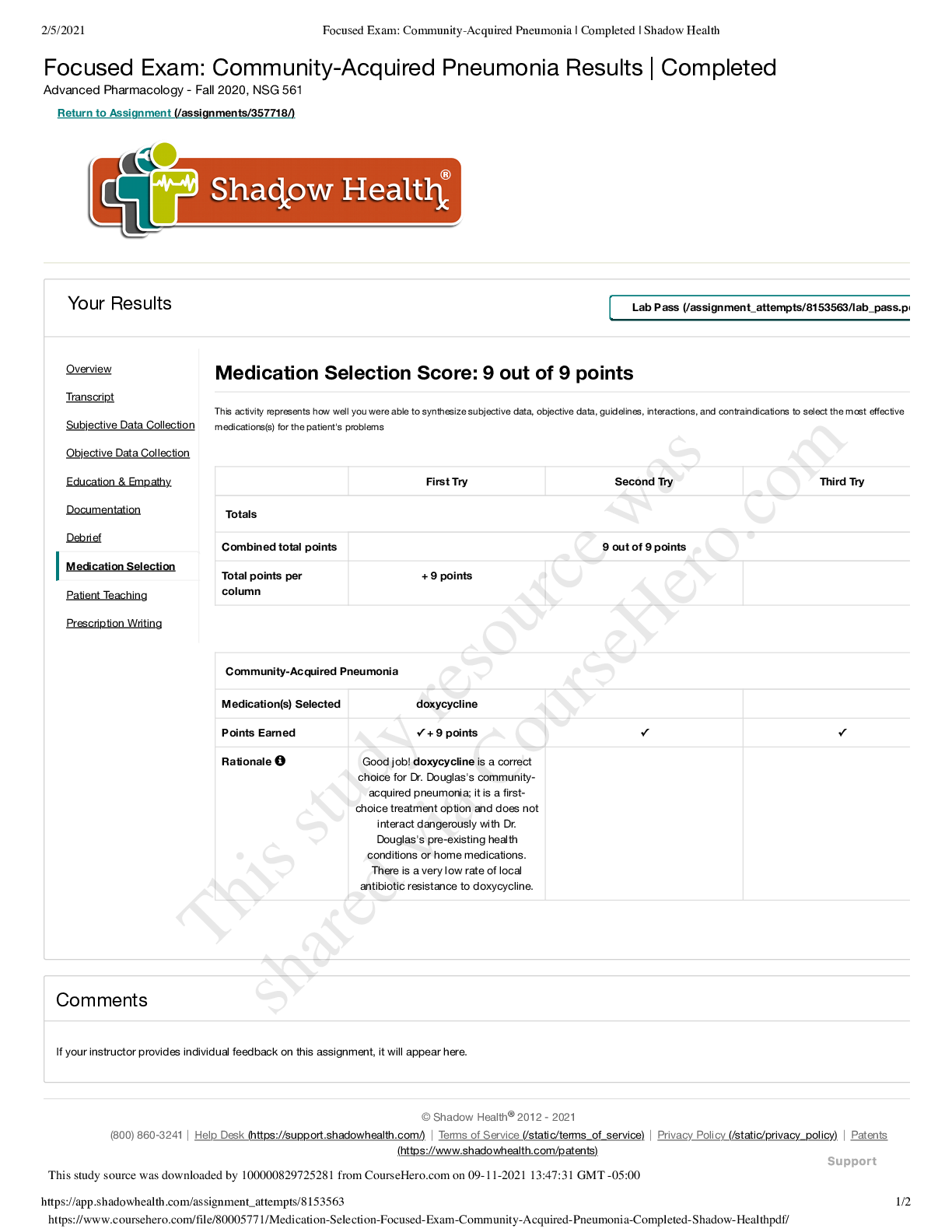
SOLVED - Elaborated SOCRA Certification Exam Final SOCRA Certification Exam Does the FDA consider electronic signatures to be as trustworthy and reliable as handwritten paper signatures? - AnsYes (a... lthough permission to use such e-sigs has to be approved by the FDA) Does the FDA consider electronic records that meet requirements to be equivalent to handwritten records ? - AnsYes Open system (FDA term) - AnsSystem access is NOT controlled by people who are responsible for the content of the electronic records in the system. (Like me putting data into CHOP - controlled databases) Closed system (FDA term) - AnsEnvironment in which SYSTEM ACCESS is controlled by the same people responsible for the content of the system (I.E. I control the Robotic Database access AND its contents) What are some FDA Standards to meet when operating a closed record system? - Ans1. Must be able to tell if records have been altered or invalid 2. Must be able to copy records for agency review 3. Protect records throughout retention period 4. Limit system to authorized individuals only 5. Use time-stamped audit trails of modification etc 6. Use operational system checks and restrictions 7. Use authority checks to make sure only authorized individuals are using the system 8. Use device checks to validate data input 9. Make sure those authorized to use system have appropriate training, education, experience 10. Have written policies that deter data falsification 11. Audit and control the maintenance of the actual system What are some FDA standards to meet when using an Open System? - AnsAll those mentioned for the closed system. 1. Document encryption as appropriate What information should a handwritten SIGNATURE block contain? - Ans1. Printed name of signer 2. Date and Time when signature was executed 3. The MEANING associated with the signature (approval? responsibility? authorship?) Signature and record linking ? - AnsSignatures must be linked to their respective electronic records to make sure they cannot be copied, falsified, transferred etc. Do researchers need to request permission from the FDA to use electronic signatures in place of regular signatures? - AnsYes. [Show More]
Last updated: 1 year ago
Preview 1 out of 14 pages
Instant download

Instant download
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Apr 20, 2023
Number of pages
14
Written in
Additional information
This document has been written for:
Uploaded
Apr 20, 2023
Downloads
0
Views
49