Astronomy > Lab Report > University of Colorado, Boulder - ASTR 1010Spectroscopy Lab II (All)
University of Colorado, Boulder - ASTR 1010Spectroscopy Lab II
Document Content and Description Below
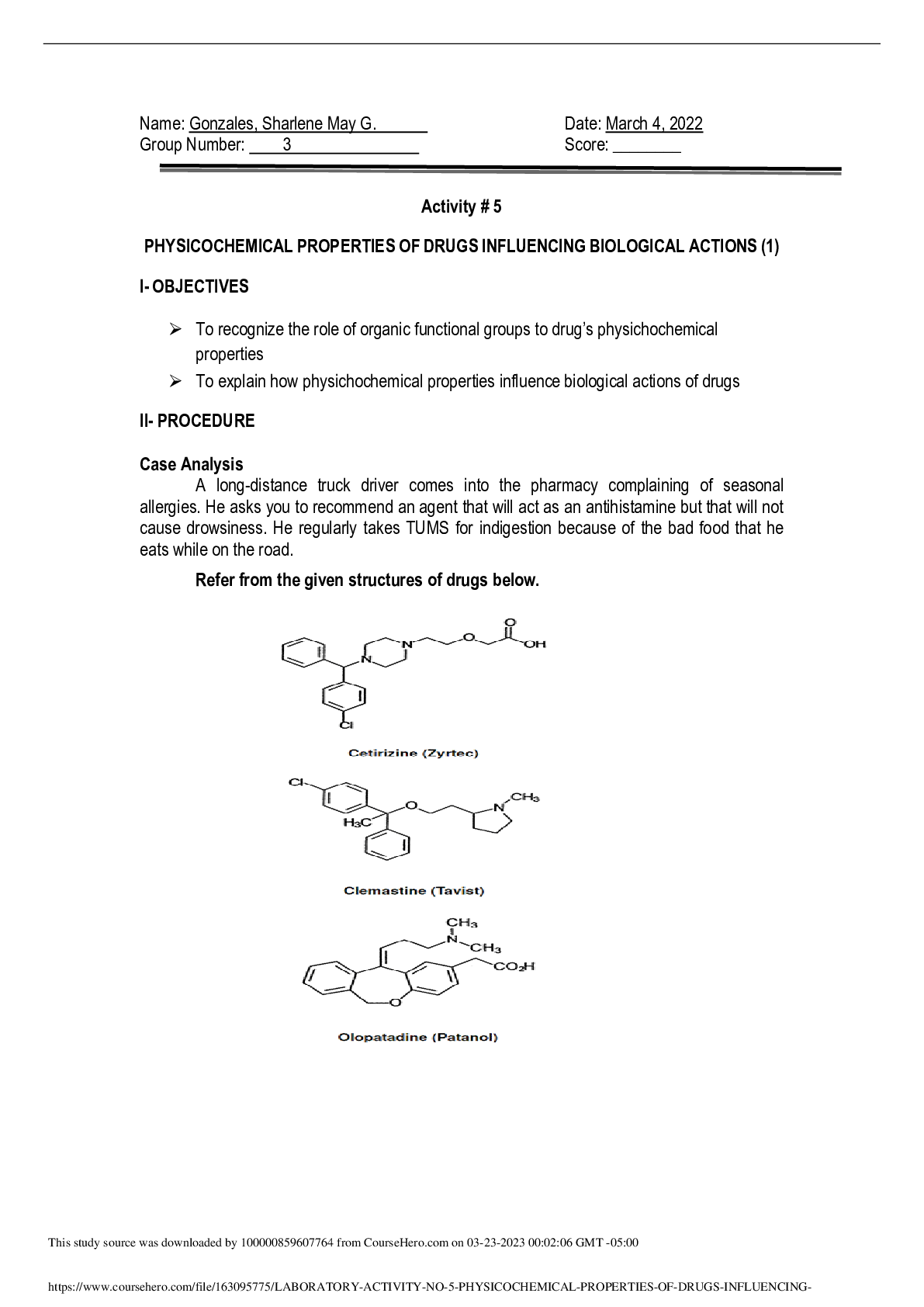
Margaret Ryan 3/21/12 ASTR 1010: Introductory Astronomy – Spectroscopy II – Spectral Barcodes Introduction: Spectroscopy is the study of colors of light emitted by objects and it is used to id... entify compositions, temperatures, velocities, pressures, and magnetic fields. The purpose of this lab was to help us understand how many objects in astronomy need to be studied from a distance by means of visible light. Also, the other learning goal is what light can tell us about the chemical composition of the object that produced the light. Part I- Electron Energy Transitions 1.) An atom can give off light by it absorbing energy, which bumps electrons into higher shell levels. When they jump back down, they emit light. a.) If an atom gives off both red and blue light it means that the electron keeps transitioning from lower levels to higher levels. 2.) An atom absorbs light when energy is put into it and the electrons jump into different levels. a.) Atoms don’t give off red or blue light. 3.) Atoms will only emit light if all of the atom’s electrons are in the ground state only if they had just moved back down to the ground state [Show More]
Last updated: 1 year ago
Preview 1 out of 2 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Mar 31, 2021
Number of pages
2
Written in
Additional information
This document has been written for:
Uploaded
Mar 31, 2021
Downloads
0
Views
82




.png)








.png)
.png)



.png)






 (1).png)