Chemistry > Lab Report > University of Colorado, Boulder - CHE 111Lab 7 - Chemical Bonding and Molecular Geometry (All)
University of Colorado, Boulder - CHE 111Lab 7 - Chemical Bonding and Molecular Geometry
Document Content and Description Below
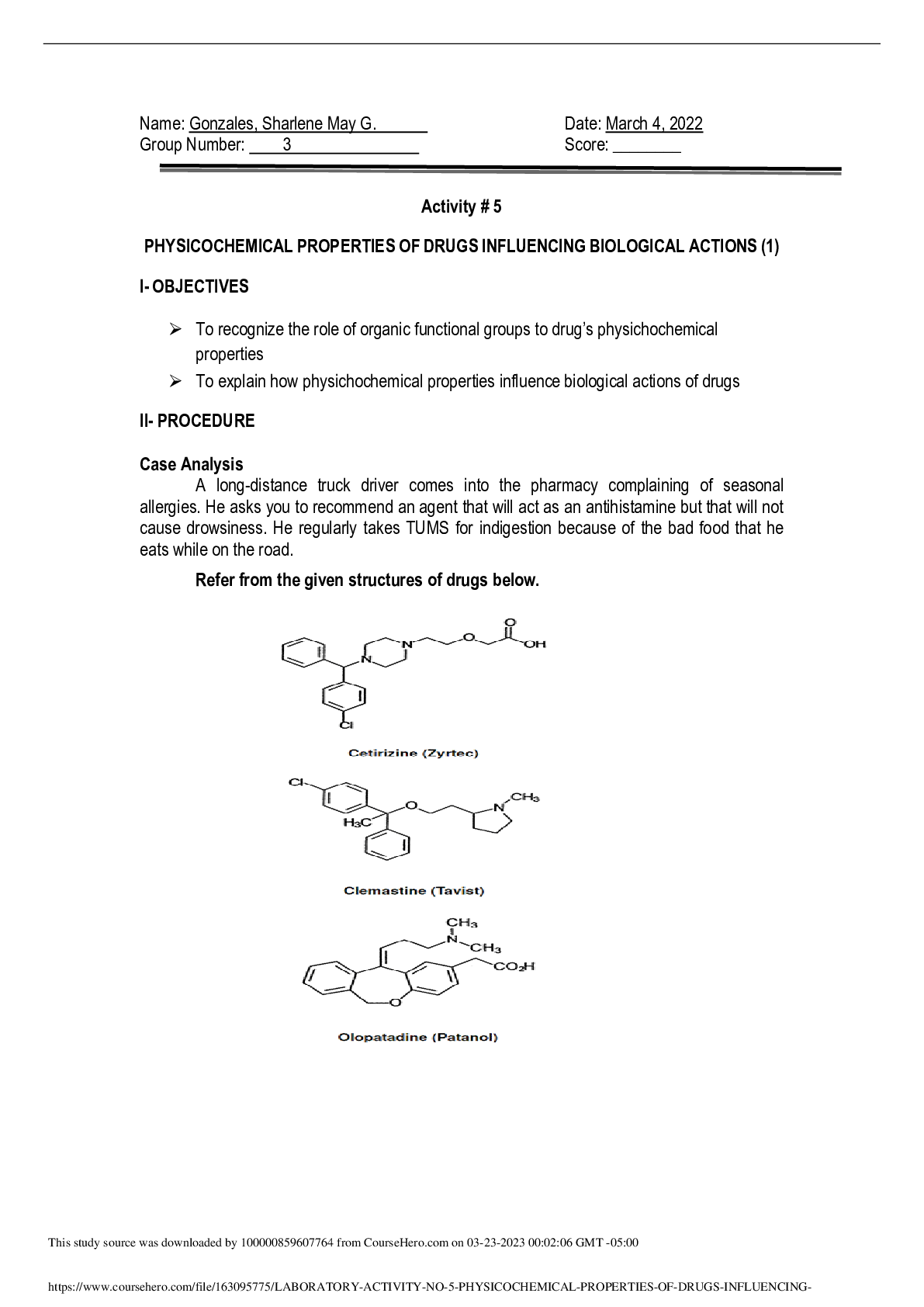
Lab 7: Chemical Bonding and Molecular Geometry 10/10/19 CHE111C03 Lab Partner: N/A Purpose: The purpose of this lab is to understand the Lewis Dot Structure as well as the VSEPR model, then appl... y them to multiple molecules. In this lab, we also work on our understanding of identifying valence electrons and the molecular geometry. Through this lab, we gain an understanding of these concepts and apply our knowledge to understand the topic. Experimental Narrative: The Lab kit was used for this lab. First, the lab manual was read to understand the procedure and materials needed. The needed materials from the lab kit were gathered and organized. The implementation of a lab coat, safety glasses, and other safety equipment were not used. First, the valence electrons of each molecule were calculated. The Lewis dot structures were drawn following the calculations of the valence electrons. The Lewis Structure bag from the lab kit was then used to reconstruct the VSEPR models of each of the molecules. This completed table 1 data. Following this, the number of atoms and lone pairs of electrons on the central Adam were noted and written within data table 2. The molecular geometry was then determined. This completed both tables and the procedure of the lab. Chemical Bonding and Molecular Geo [Show More]
Last updated: 1 year ago
Preview 1 out of 13 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Apr 21, 2021
Number of pages
13
Written in
Additional information
This document has been written for:
Uploaded
Apr 21, 2021
Downloads
1
Views
134

.png)


.png)


.png)



.png)









 (1).png)