Physics > Lab Report > Course Activity_ Combining Objects at Different Temperatures>Determining Heat Gain and Loss lab repo (All)
Course Activity_ Combining Objects at Different Temperatures>Determining Heat Gain and Loss lab report
Document Content and Description Below
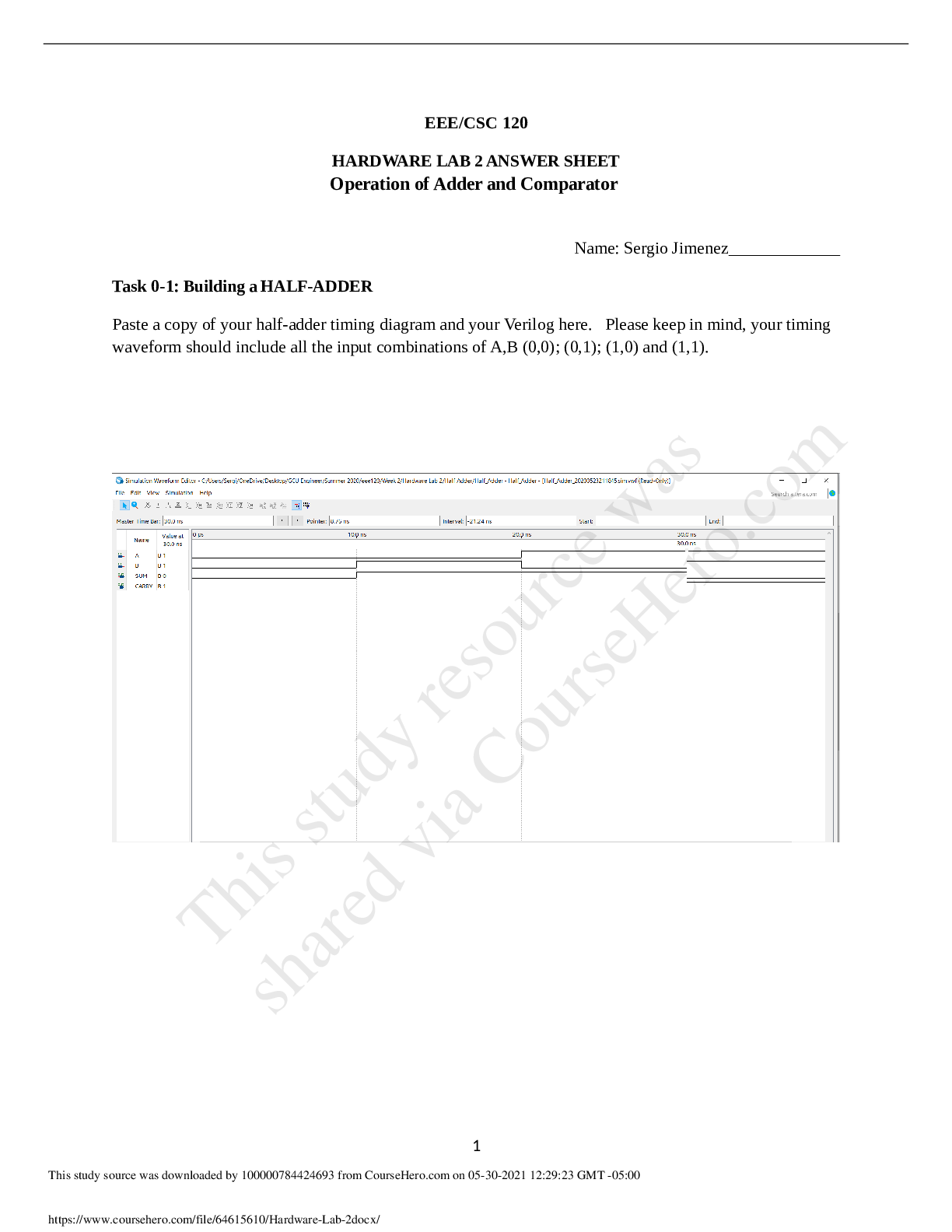
Determining Heat Gain and Loss In this activity, you’ll heat an object in hot water and then transfer it to cool water. The object will become cooler, and the cool water will become warmer. Based ... on the temperature changes, you’ll calculate and compare the heat lost by the object and the heat gained by the water. To begin, choose an object (or objects) made of a known material and that can be immersed in water without causing damage. Before you begin this task, read these lab safety guidelines . If you’re using an electronic balance, watch this video about measuring mass . Estimated time to complete: 1 hour If you’ve purchased an Edmentum lab kit, remove the electronic balance and thermometer from the kit. These materials are italicized in the following equipment list. Then gather any additional items shown in the list. If you’re not using an Edmentum lab kit, alternatives are suggested in parentheses. You’ll need these materials: electronic balance (may also use a kitchen scale, readable at least to the nearest gram) thermometer, readable at least to the nearest degree from 0°C to 100°C (32°F to 212°F)—readings to the nearest tenth of a degree will produce better results foam cups (2) lids to Õt the cups (may use squares of cardboard as lids) solid object (or group of objects) of a known material that can Õt easily in the cup, ideally having a total mass of 25 to 60 grams; here are some ideas: 2–5 plastic dominoes, depending on size 5–10 glass marbles, depending on size 10 quarters tongs pen or marker stove, hot plate, or microwave oven container for heating water (depends on the selected heat source) Part A Begin the task by performing these steps. Record your observations in the table provided. Label the foam cups with the numbers 1 and 2. Record the type (and quantity, if applicable) of object you will be using. Include what kind of material the object is made of. mDetermine the speciÕc heat capacity of the chosen object from this list of speciÕc heat capacities . If you can’t locate your material on the list, do an online search. Use the electronic balance to measure the mass of the chosen object(s). Record the mass. Then, place the object(s) into cup 1. Answer: Name of object and quantity 6 balls Material aluminum SpeciÕc heat capacity (J/(g °C)) 0.91 Total mass (g) 50,6 Characters used: 115 / 15000 Part B Measure and record the mass of cup 2, empty and without the lid. Put cold or room-temperature water into cup 2. There should be at least enough water to completely cover the object(s) once it’s moved from cup 1. For best results, the cup shouldn’t be completely full. Measure and record the mass of cup 2 with water in it, but without the lid. Calculate the mass of the water in the cup. [Show More]
Last updated: 1 year ago
Preview 1 out of 7 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Apr 16, 2021
Number of pages
7
Written in
Additional information
This document has been written for:
Uploaded
Apr 16, 2021
Downloads
2
Views
255

.png)



















 (1).png)






.png)


.png)

