Chemistry > LECTURE NOTES > Carbon and its Compounds (All)
Carbon and its Compounds
Document Content and Description Below
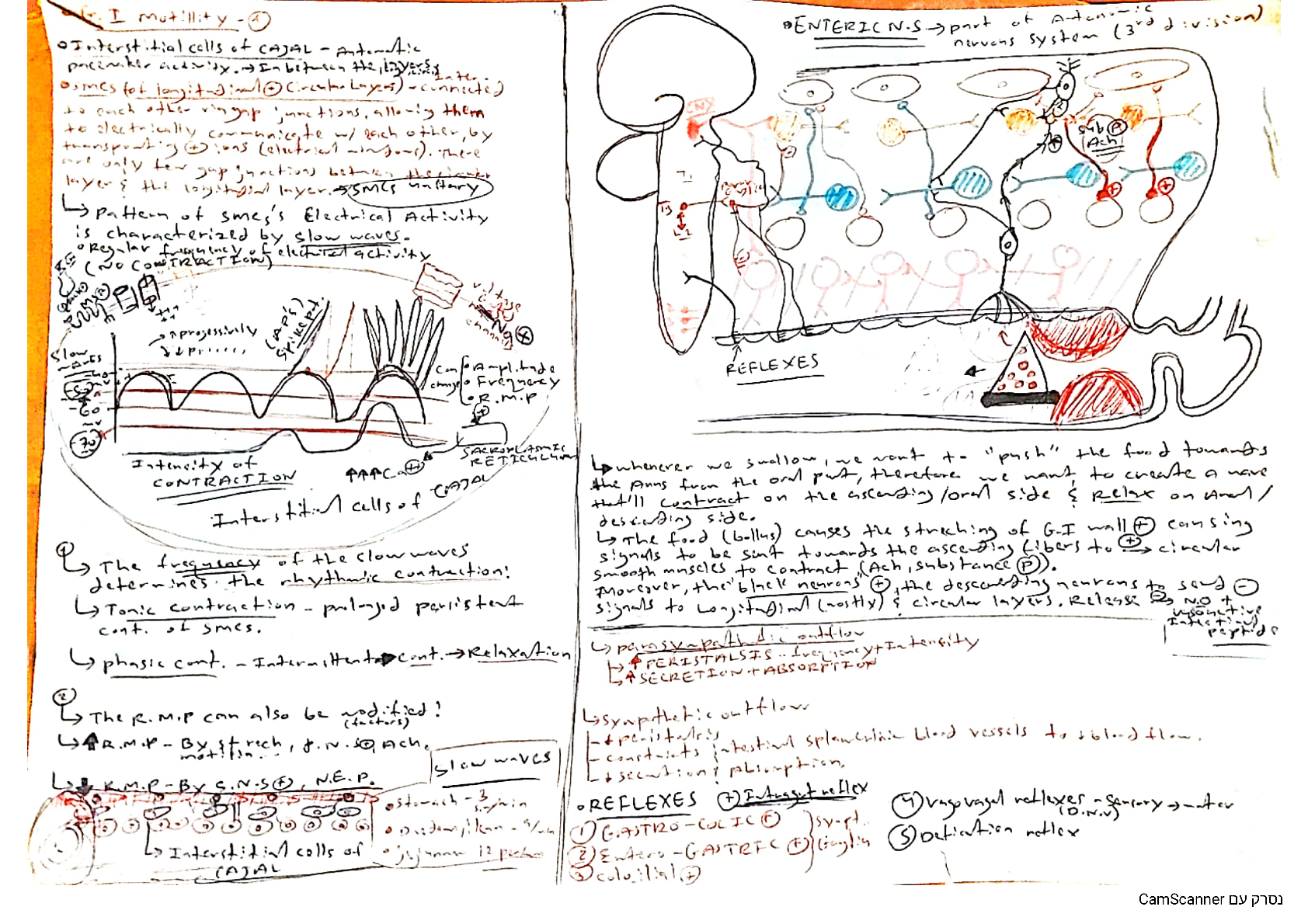
Why Carbon forms Covalent Bonds We know that a majority of substances used by us daily, from paper and plastics to coal and petrol, are all made up of carbon. Food grains, pulses, medicines, cotton,... synthetic fibres, wood etc. are all made up of carbon. Carbon is also a major part of all living things. In air, it is present as carbon dioxide and comprises around 0.03% of the total air. Let us study about carbon and its bonding in its compound in more detail. Carbon is a non-metal having the symbol ‘C’ and atomic number six. Since the atomic number of carbon is six, its electronic configuration is 2, 4. This means that carbon contains two electrons in K shell and 4 electrons in L shell (outermost shell). Hence, it has four electrons in its valence shell. Since carbon has four electrons in its valence shell, it needs four more electrons to complete its octet. Therefore, it is a tetravalent element. [Show More]
Last updated: 1 year ago
Preview 1 out of 38 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Aug 12, 2021
Number of pages
38
Written in
Additional information
This document has been written for:
Uploaded
Aug 12, 2021
Downloads
0
Views
64






.png)












