CHEM 120 Unit 3 Quiz Graded with all answers correct
Document Content and Description Below
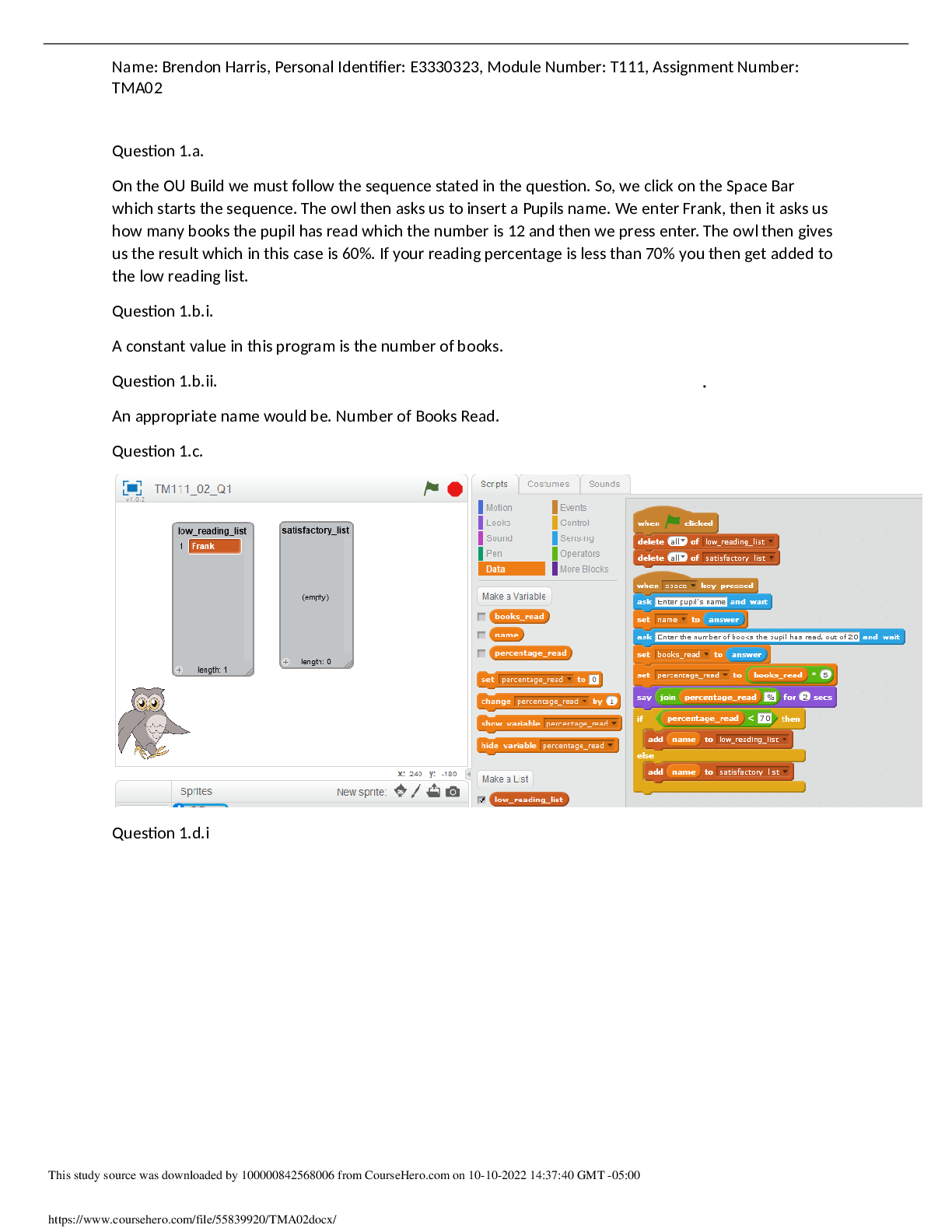
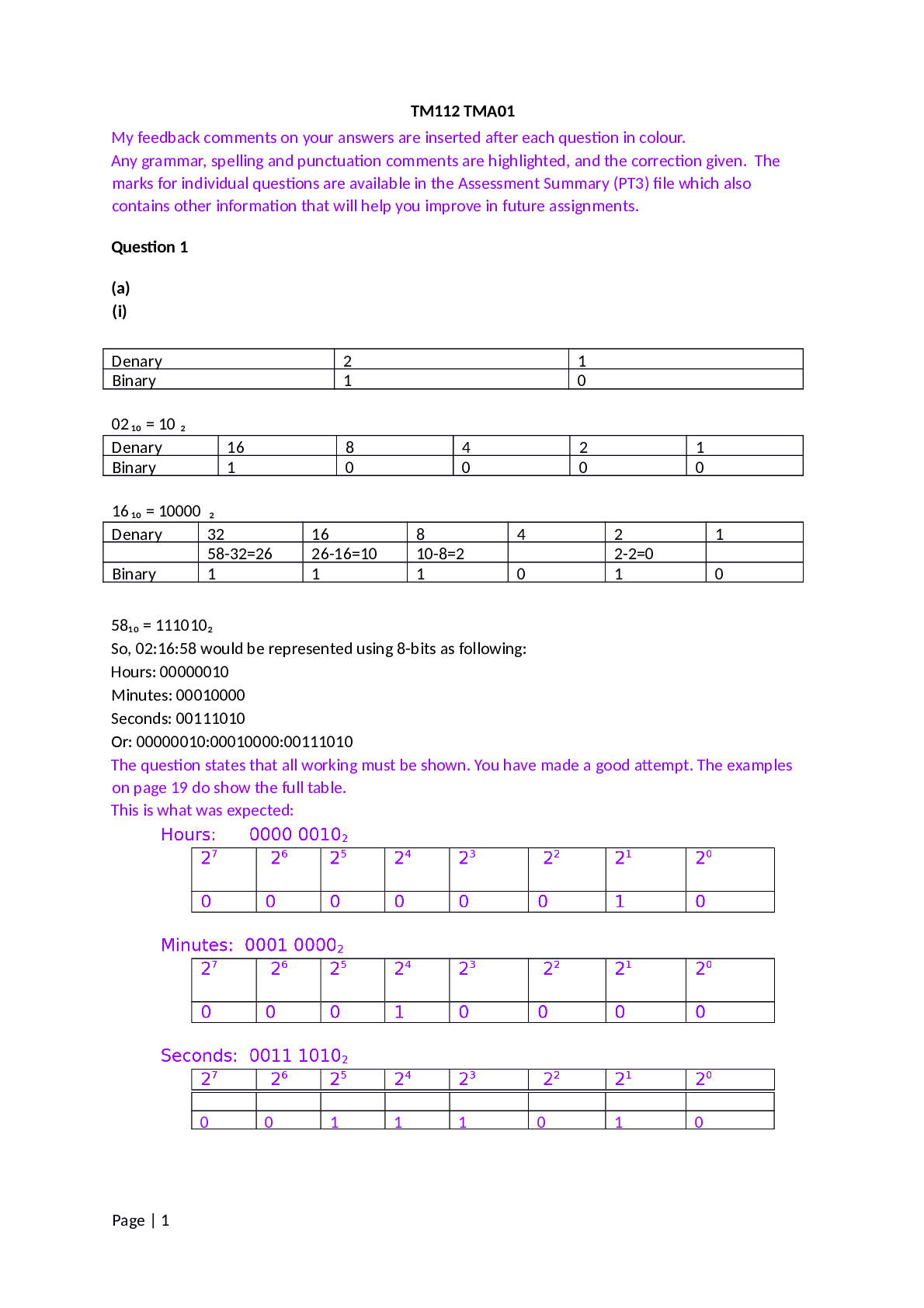
Question: A reaction that releases energy as it is classified as an ____ while a reaction that absorbs energy as it occurs is classified as an ____. Question:Which coefficient is placed in front of p... otassium to balance the following equation? K+H2SO4-K2SO4+H2= ____ Question: Which coefficient is placed in front of HCI to balance the following equation? Ca(OH)2 +? HCICaCl2 +H2O = ____ Question: Sodium reacts with oxygen to form sodium oxide and has the following chemical equation: 4 Na + O2 How many mole(s) of oxygen gas (O2) are needed to react with 8.0 moles of Na? 2.0 Question: Magneisum reacts with oxygen to form magnesium oxide and has the balanced chemical equation: 2Mg +O2 2 MgO. How many moles of MgO are produced when 5 moles O2 reacts completely? Question: Determine number of atoms of the following molecule Na2CO3. Using periodic table determine the molar mass for this molecule: Question: Determine number of atoms of the following molecule BaSO4. Using periodic table determine the molar mass for this molecule: Question: Calculate the concentration of NaCl (in water) solution in several ways and are given the following info: 25g NaCl, 500mL solution. Molecular weight of NaCl= 58.44 calculate mass/volume percent concentration and molarity of the solution Question: What volume (L) of a 1.5 M KOH solution can … prepared by diluting0.2 L of a 5M KOH solution? Question: What is the molecular mass of N2O [Show More]
Last updated: 1 year ago
Preview 1 out of 1 pages
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Jan 26, 2021
Number of pages
1
Written in
Additional information
This document has been written for:
Uploaded
Jan 26, 2021
Downloads
0
Views
42






.png)