2024 CPPS Exam Review ( Notes ) Complete Answers 100% Correct
Document Content and Description Below
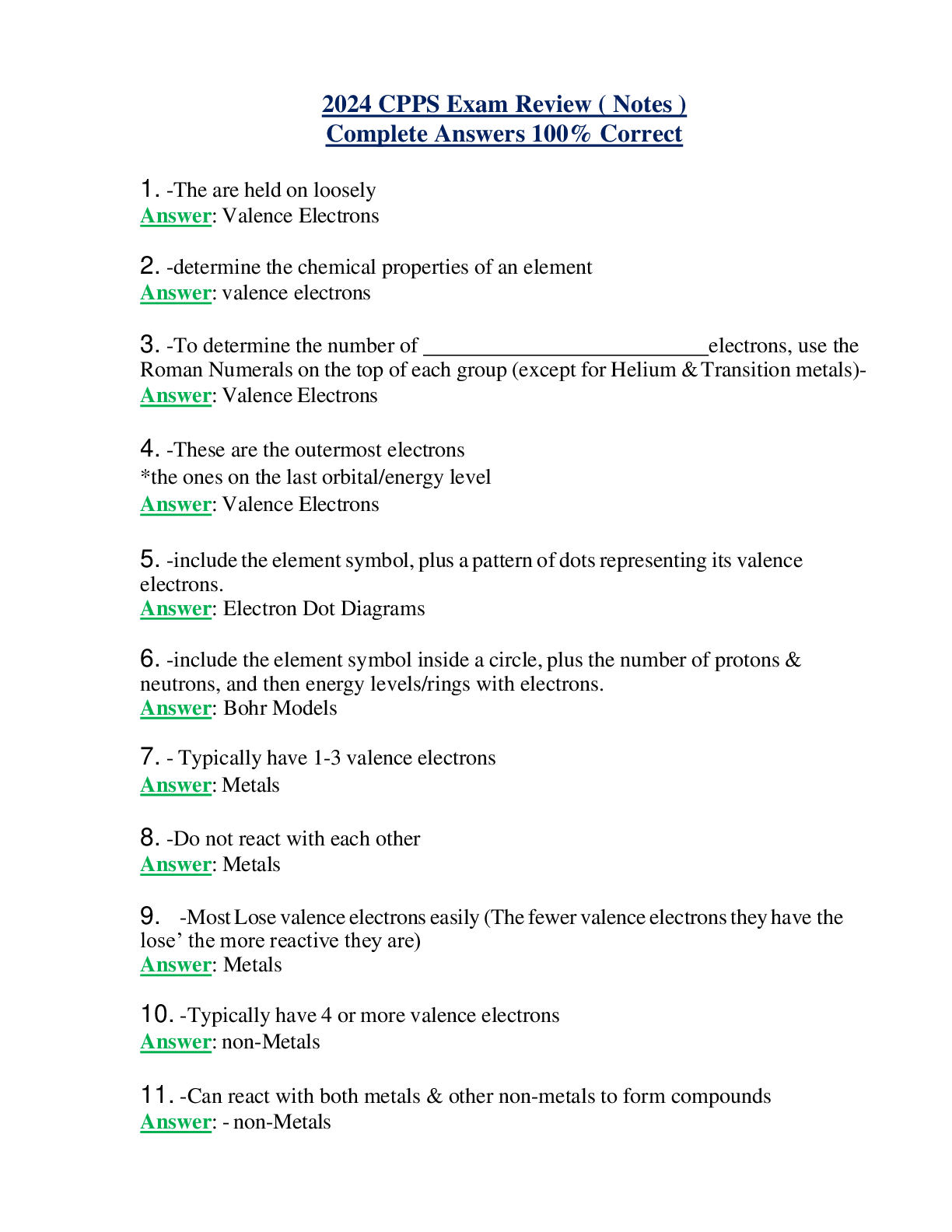
1. -The are held on loosely Answer: Valence Electrons 2. -determine the chemical properties of an element Answer: valence electrons 3. -To determine the number of electrons, use the Roman Numera... ls on the top of each group (except for Helium &Transition metals)- Answer: Valence Electrons 4. -These are the outermost electrons *the ones on the last orbital/energy level Answer: Valence Electrons 5. -include the elementsymbol, plus a pattern of dotsrepresenting its valence electrons. Answer: Electron Dot Diagrams 6. -include the element symbol inside a circle, plus the number of protons & neutrons, and then energy levels/rings with electrons. Answer: Bohr Models 7. - Typically have 1-3 valence electrons Answer: Metals 8. -Do not react with each other Answer: Metals 9. -MostLose valence electrons easily (The fewer valence electronsthey have the lose’ the more reactive they are) Answer: Metals 10. -Typically have 4 or more valence electrons Answer: non-Metals 11. -Can react with both metals & other non-metals to form compounds Answer: - non-Metals [Show More]
Last updated: 2 months ago
Preview 1 out of 23 pages
Instant download
Buy this document to get the full access instantly
Instant Download Access after purchase
Add to cartInstant download
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Apr 15, 2024
Number of pages
23
Written in
Additional information
This document has been written for:
Uploaded
Apr 15, 2024
Downloads
0
Views
21