Pharmacology > QUESTIONS & ANSWERS > MPJE Questions and Answers Rated A+ (All)
MPJE Questions and Answers Rated A+
Document Content and Description Below
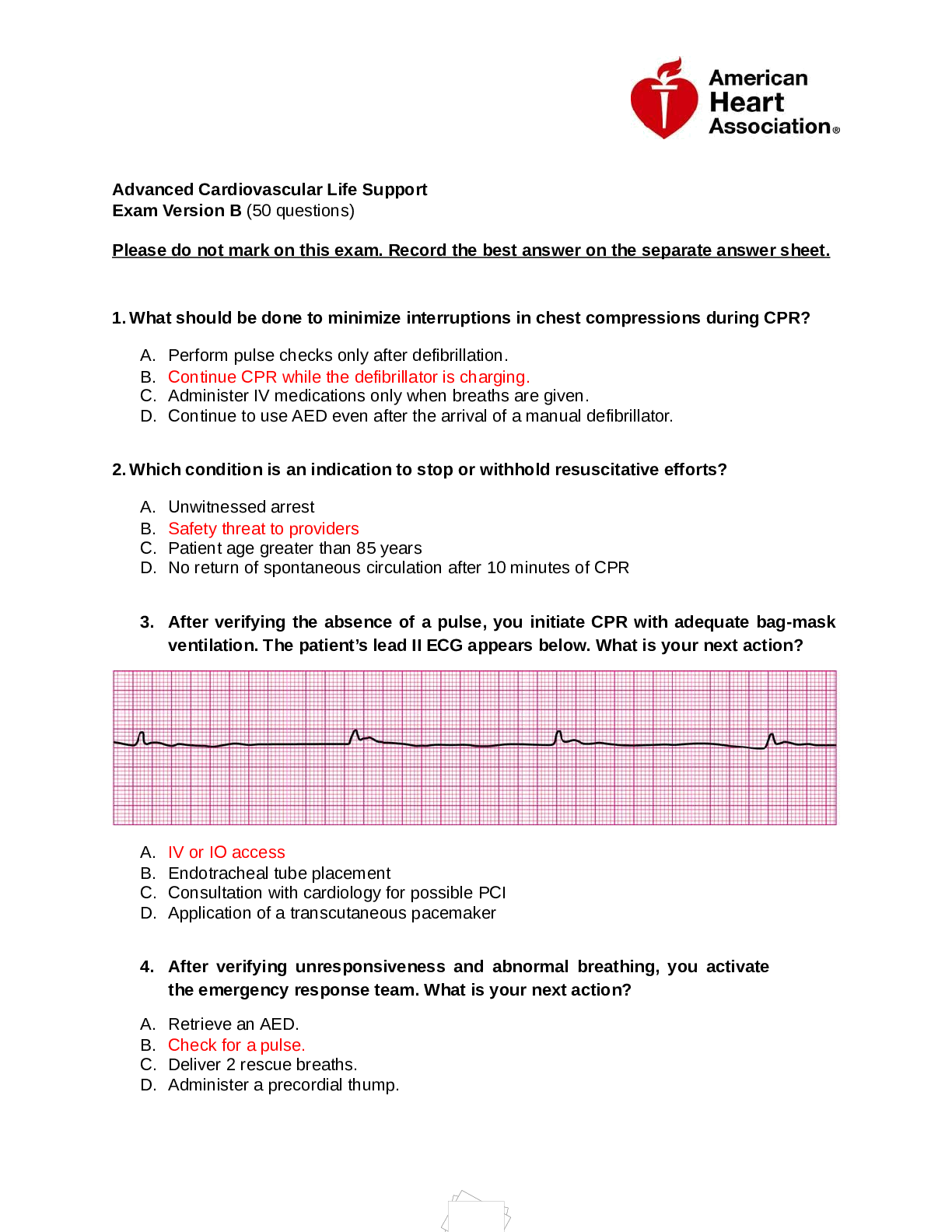
MPJE Questions and Answers Rated A+ DEAform 224 renew every? ✔✔new pharmacy registration to dispense controlled substances (application for new registration) - pharmacy registration is renewed ... every 3 years (renewal registration form DEA form 224a - chain=224b) - registration kept onsite - issued by attorney gen. 3 exceptions for registration for dispensing controlled substances ✔✔employees, contract carrier, ultimate user who has to obtain a registration as a chemical distributor? who is exempt? what are the drugs listed here? ✔✔-wholesale distribution -retail pharmacies registered for controlled substances and retail distributors who sales are for personal use are below threshold quantities -ephedrine,pseudoephedrine, phenylprolanolamine (PPA), and ephedrine combos (List 1 chemicals) not to exceed 3.6g/day or 9g/month DEA form 510 ✔✔DEA chemical distributor registration the attorney general can suspend or revoke DEA registration with what findings ✔✔falsified application, felony conviction involving controlled substances or list 1 chemicals, license suspended,revoked or denied, excluded from medicare/medicaid, act inconsistant with public interest 4 ways to get rid of controlled substances records kept for how long ✔✔-transfer to customer, dispensing rx -destruction -transfer to another DEA registrant (dr,back to distributor or manufacturer) -surrender to DEA/law enforcement >2 years, readily available (5yrs UCSA) how to transfer C2 substances to another DEA registrant, pharmacy, manuf? C3-C5? five percent rule? ✔✔receiving registrant must issue Form 222 (dr brings 222 form into the pharmacy) - also need if transfering ownership of pharmacy w/ inventory- notify DEA of transfer 14 PT - keep records for 2 years -just need documentation with drug name, dosage form, strength, quant, date and name, address, DEA number of those involved note:if transfering >5% of yearly sold controlled than need to register as a distributor!!! (five percent rule) DEA form 41 ✔✔form submitted to the special agent in charge (3 copies) that list controlled substances desiring to dispose of - form needed to report disposal or breakage/spillage 4 ways controlled substances can be destroyed ✔✔auth by special agent to do one of the following -transfer to person registered -delivery to an agent -destruction in presence of an agent -another means determined by agent (per UCSA:maintain records for 5 yrs) DEA states schedule 2,3,4,5 should be... ✔✔in a locked cabinet or dispersed in non-controlled stock to deter theft. electronic alarm recommended [Show More]
Last updated: 1 year ago
Preview 1 out of 92 pages
.png)
Also available in bundle (1)
.png)
MPJE BUNDLED EXAMS QUESTIONS AND ANSWERS WITH VERIFIED SOLUTIONS
MPJE BUNDLED EXAMS QUESTIONS AND ANSWERS WITH VERIFIED SOLUTIONS
By Nutmegs 1 year ago
$27
23
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Mar 20, 2023
Number of pages
92
Written in
Additional information
This document has been written for:
Uploaded
Mar 20, 2023
Downloads
0
Views
86

.png)
.png)
.png)
.png)
.png)
.png)









.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)



.png)
.png)

.png)

.png)
.png)

