Biology > QUESTIONS & ANSWERS > University of PennsylvaniaBIOL 204BIOL 204 (2017) Problem Set 5 + Key- GRADED A. (All)
University of PennsylvaniaBIOL 204BIOL 204 (2017) Problem Set 5 + Key- GRADED A.
Document Content and Description Below
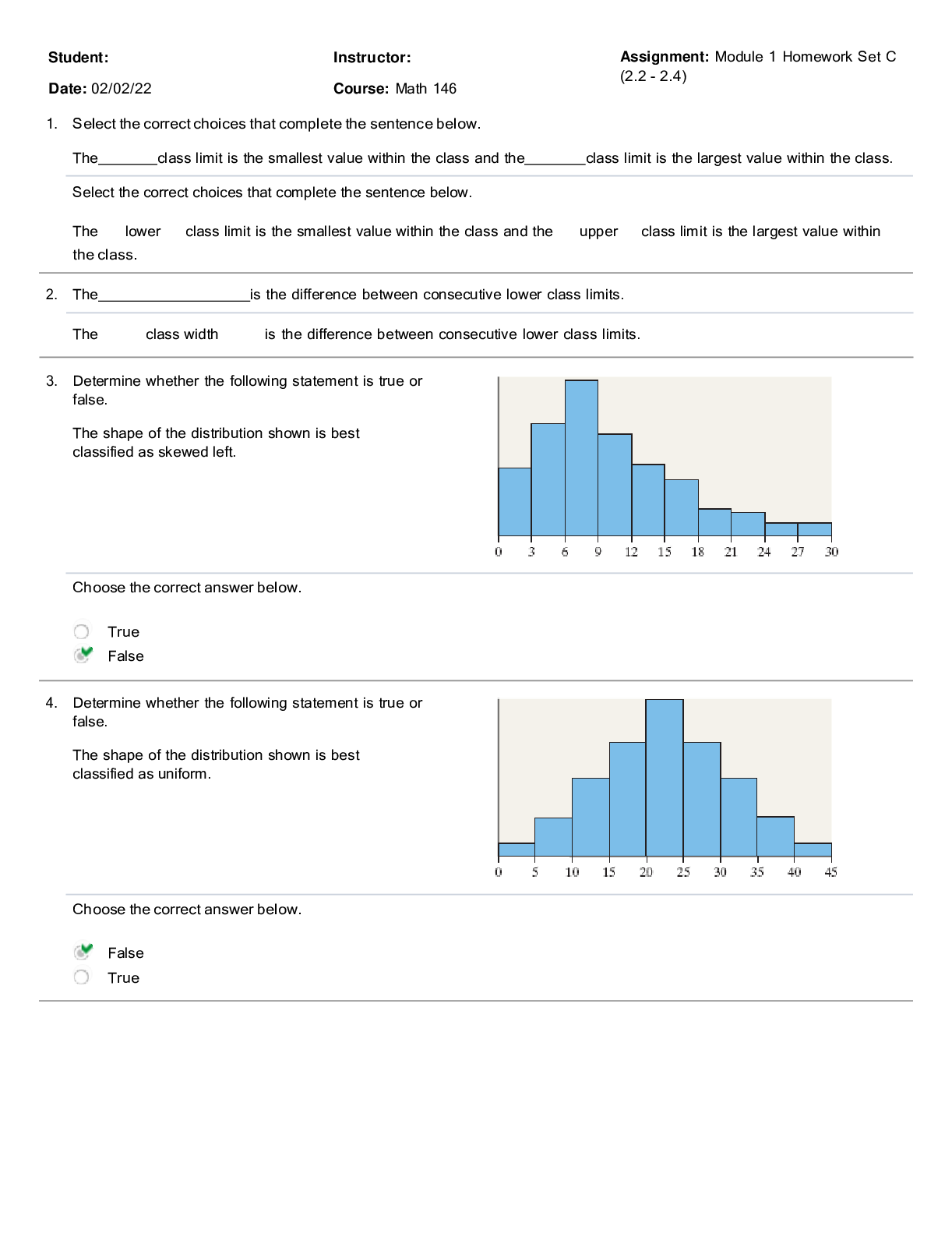
Biology 204 Spring 2020/REA/ELLIOTT 1 Problem Set 5: Intermediary metabolism Questions: These questions are intended as a supplement and complement to the material covered in the lecture series ... ‘Metabolism’ but largely excluding β-oxidation and oxidative phosphorylation which will be dealt with in Problem Set 6. In order to maximize the learning experience, it is important that you understand the problems and work on them before coming to the discussion. Take your time with these questions and do not jump to the conclusion that you do not know enough to answer them. Make full use of the course textbook and your lecture notes. 1. For the reaction A ⇌ P, K’eq(AP) = 4 x 10-4 at 37oC. For the reaction P ⇌ Q, K’eq(BQ) = 2 x 105 at 37oC. A. Determine K’eq(AQ) for the overall reaction A ⇌ Q. B. Determine the standard free-energy changes for the conversion of A to P, P to Q, and A to Q. 2. The K’ eq for the pyruvate kinase-catalyzed phosphorylation of ADP to ATP, one of the two substrate-level phosphorylation reactions of glycolysis, is 3.6 x 105 at 25oC. If the ΔG’o for the hydrolysis of ATP to ADP and Pi at 25oC is -30.5 kJ/mol, what is the free-energy of PEP hydrolysis to pyruvate and Pi under the same conditions? 3. The steady-state concentrations of ATP, ADP and Pi in the cytosol of a metabolically active E. coli cell are 7.90 mM, 1.04 mM, and 7.90 mM, respectively. Assume a standard free-energy of ATP hydrolysis (ΔG’o) of -30.5 kJ/mol A. What is the free-energy of ATP hydrolysis under these conditions? Assume a cytosolic pH of 7 and incubation temperature of 37oC. B. If the cytosolic concentration of ATP were to decrease from 7.90 mM to 0.5 mM concomitant with an increase in the concentration of ADP from 1.04 mM to 8.44 mM and of Pi from 7.90 mM to 15.30 mM, how would the free-energy of ATP hydrolysis be affected? Again, assume a cytosolic pH of 7 and incubation temperature of 37oC. C. Comment on the results of your calculations in terms of the energetic consequences of cellular ATP depletion in terms of the principle that cellular ATP concentration is generally a closely guarded parameter. 4. In the citric acid cycle, malate is dehydrogenated to oxaloacetate in a highly endergonic reaction with a ΔG’o of +30 kJ mol-1: L-malate + NAD+ ⇌ oxaloacetate + NADH + H+ A. Calculate the equilibrium constant K’eq of this reaction. What is the implication of this result?Biology 204 Spring 2017/REA/ELLIOTT 2 B. Let us suppose that you have gathered two sets of data concerning the malate dehydrogenase reaction: (a) From one set of data obtained from three different model systems (rat, mouse and pig) you have determined that the concentration of L-malate in the liver is about 0.25 mM when the [NAD+]/[NADH] ratio is approximately 10 under standard conditions (pH 7 and 25oC); (b) From another set of data obtained from mitochondria isolated from mice you have determined that oxaloacetate concentration ranges from 3-16 nM under the same standard conditions (pH 7 and 25oC) regardless of whether the mice are maintained on high or low glucose diets. Based on the first set of data, (a), calculate the concentration of oxaloacetate at equilibrium. Compare your result with the data in (b). What is the likely significance [Show More]
Last updated: 1 year ago
Preview 1 out of 13 pages
.png)
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
May 01, 2021
Number of pages
13
Written in
Additional information
This document has been written for:
Uploaded
May 01, 2021
Downloads
0
Views
38





.png)
.png)






.png)
 Problem Set 4.png)



.png)


.png)


.png)


.png)

.png)
.png)

