Chemistry > GIZMOS > Gizmo Boyle & Charles Law Student Lab Sheet Question And Answers( Download To Score An A) (All)
Gizmo Boyle & Charles Law Student Lab Sheet Question And Answers( Download To Score An A)
Document Content and Description Below
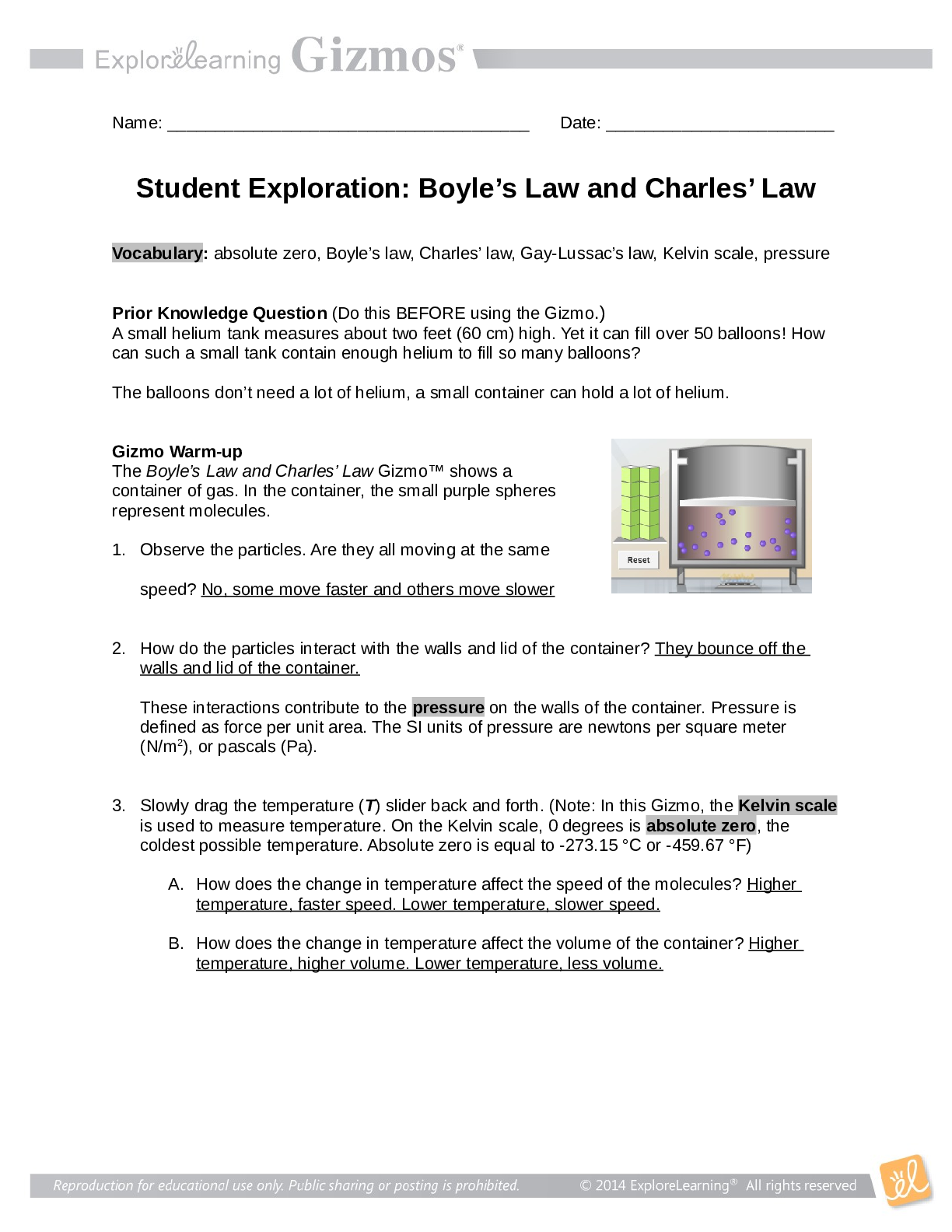
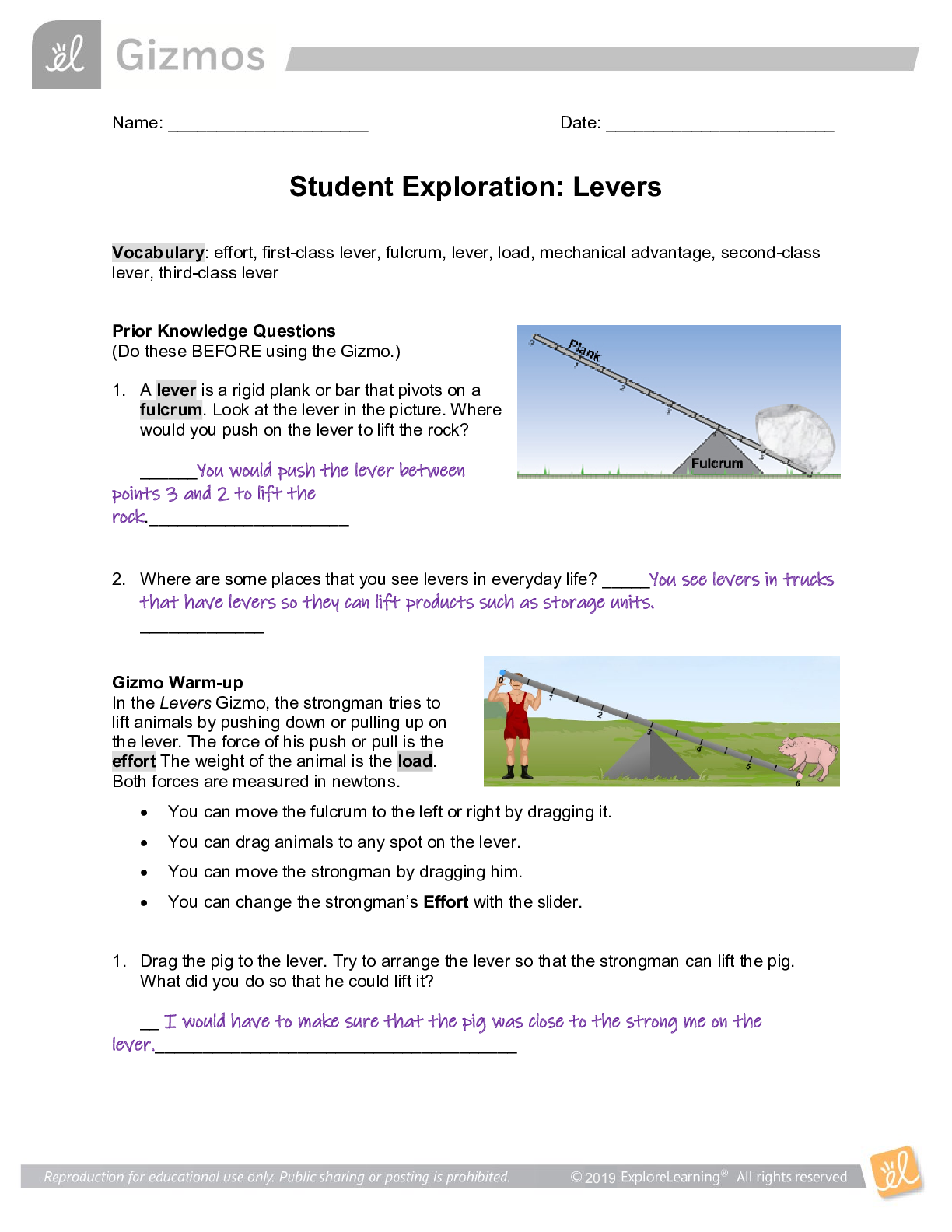
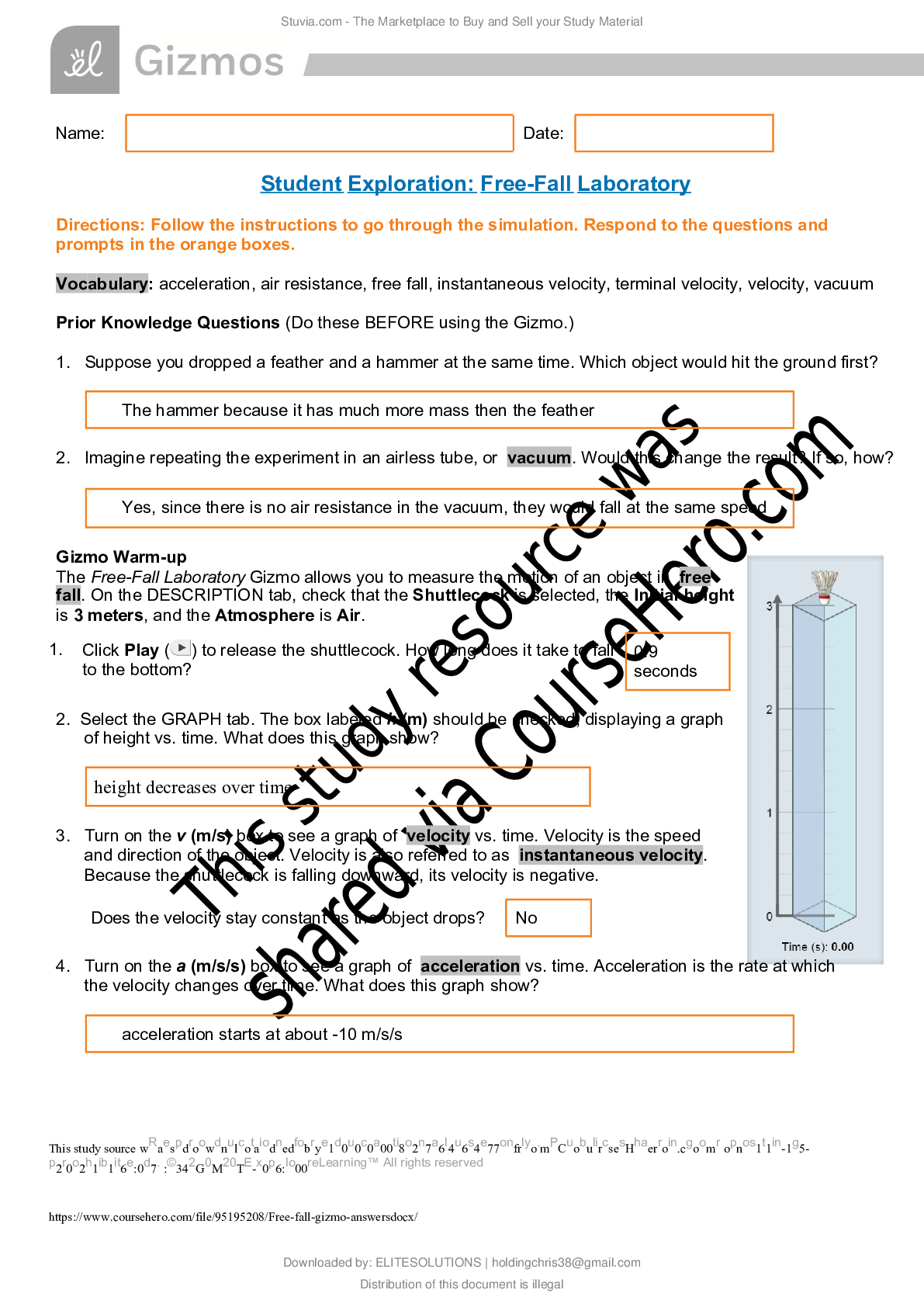
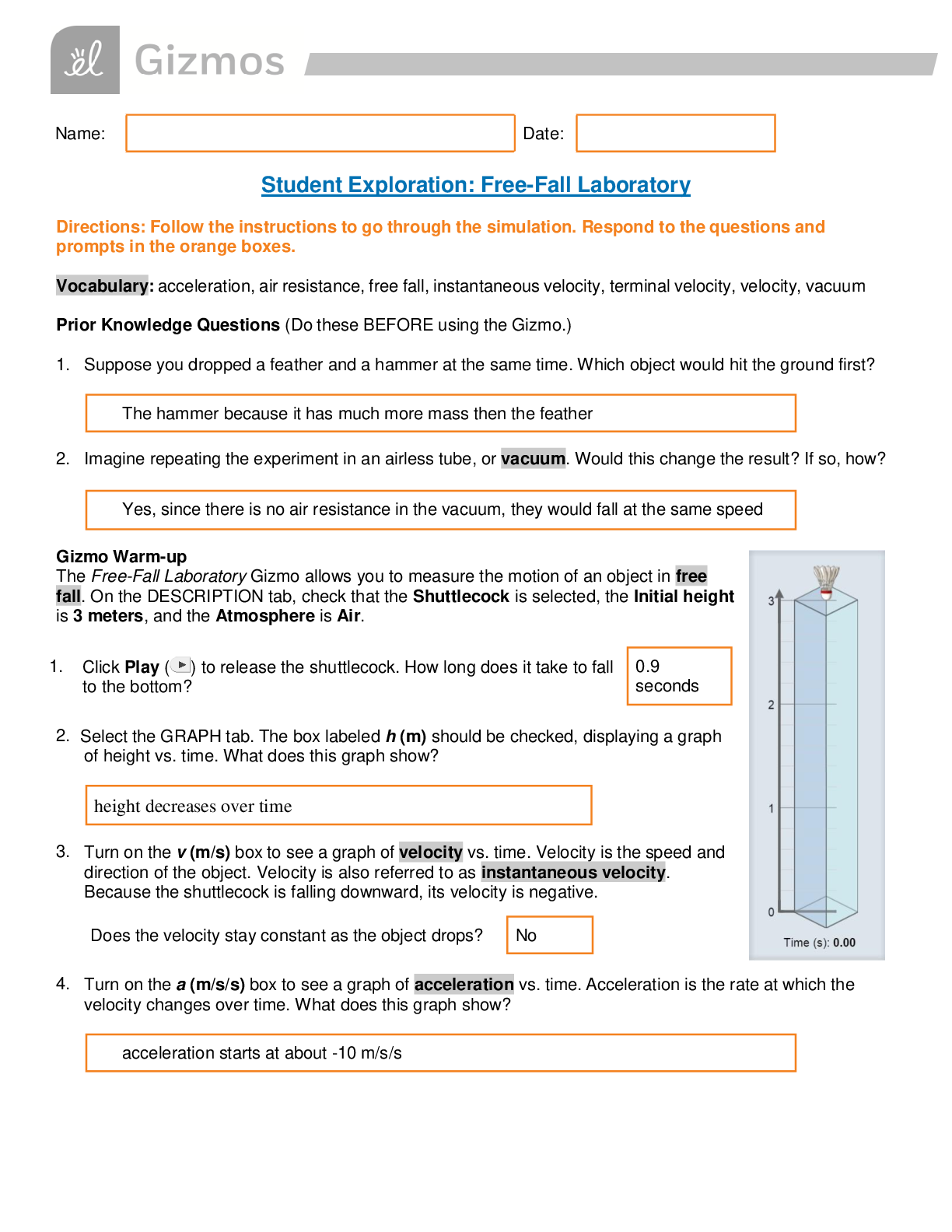
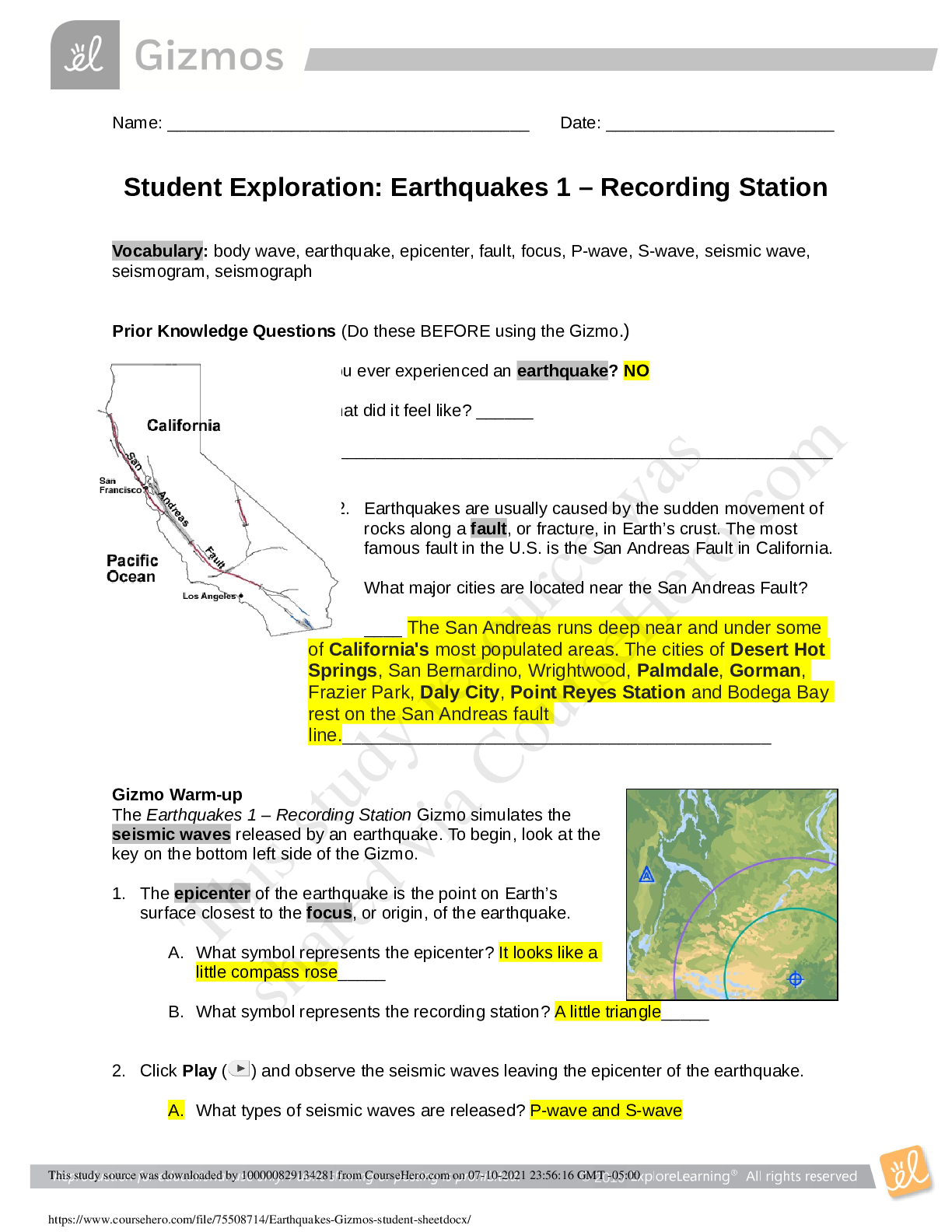
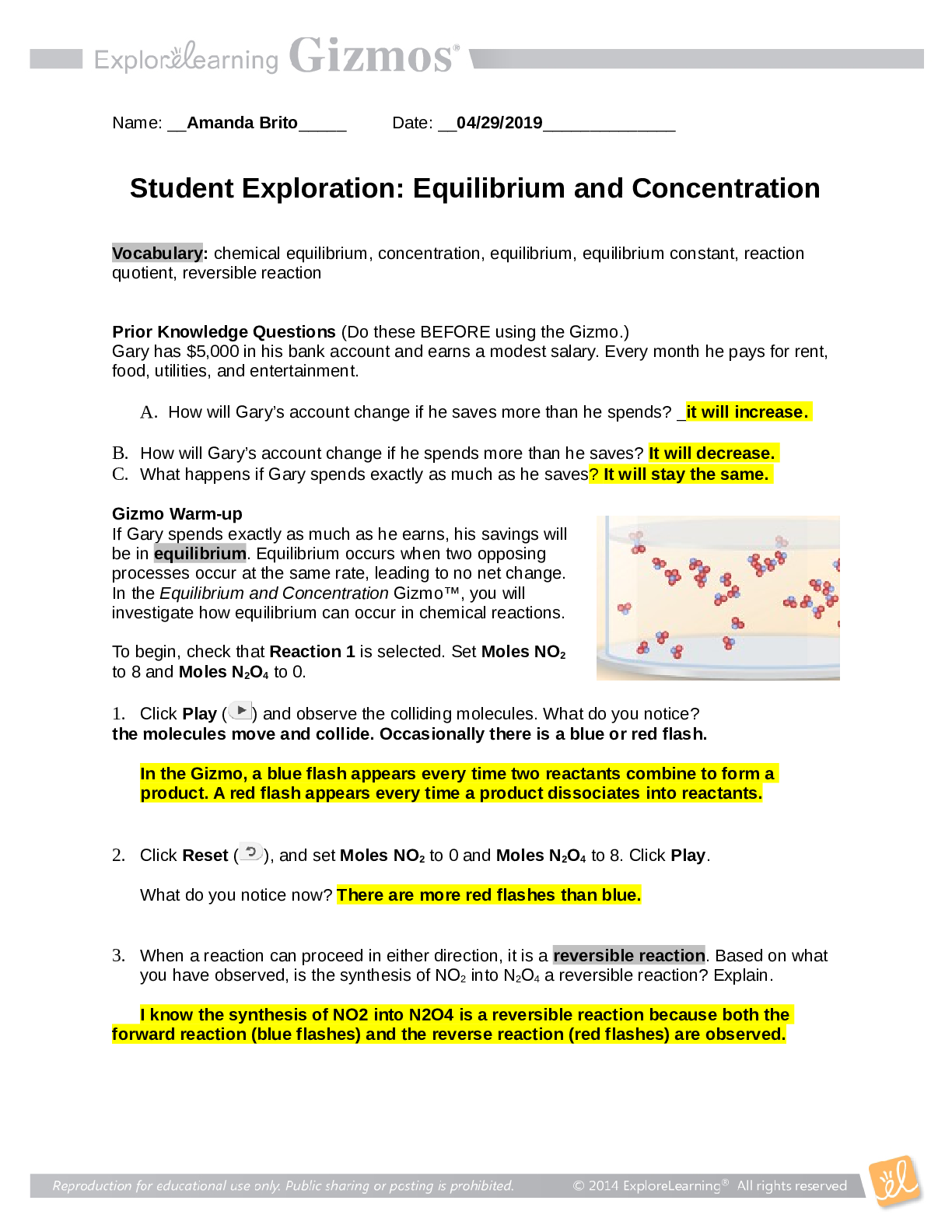
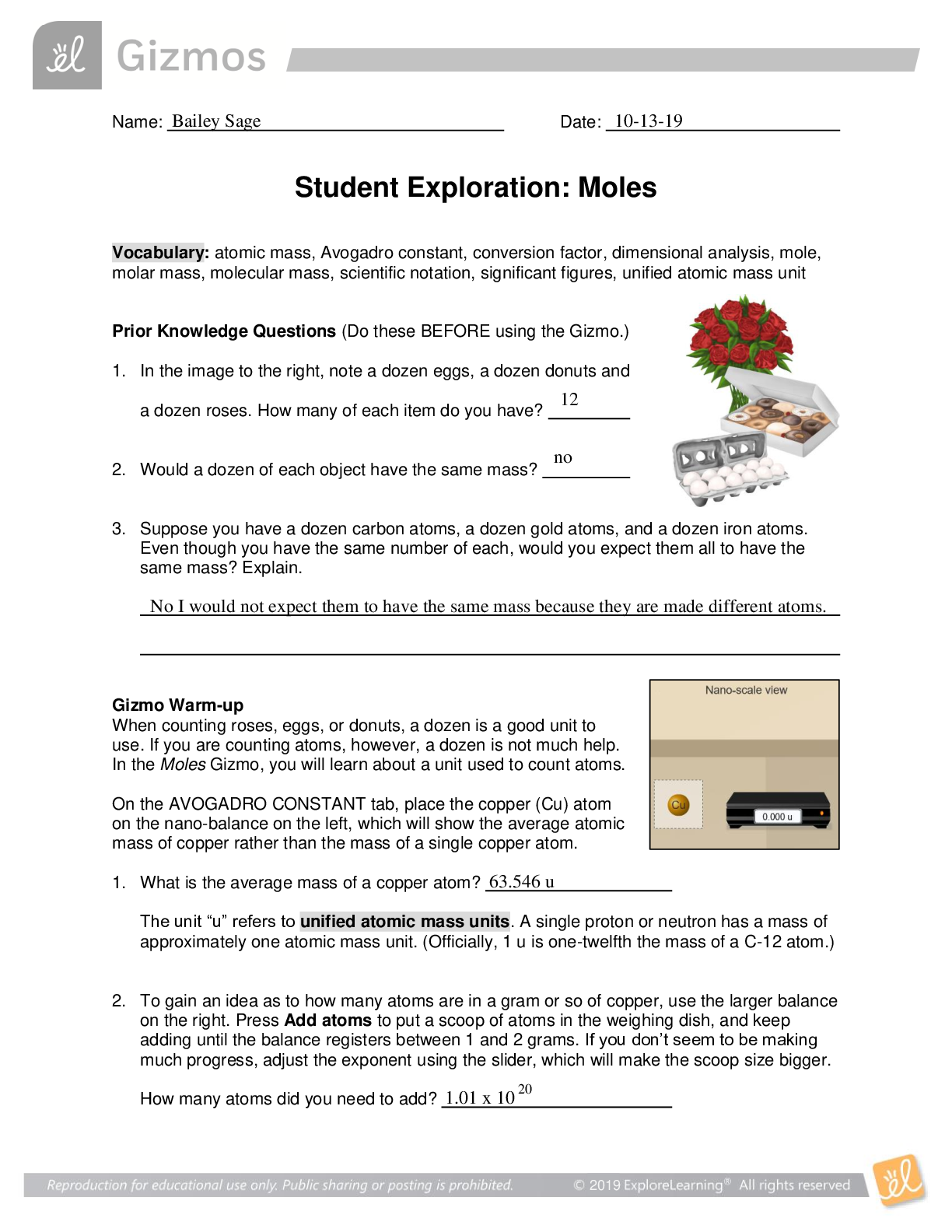
Name: ______________________________________ Date: ________________________ Student Exploration: Boyle’s Law and Charles’ Law Vocabulary: absolute zero, Boyle’s law, Charles’ law, Gay-Lussac... ’s law, Kelvin scale, pressure Prior Knowledge Question (Do this BEFORE using the Gizmo.) A small helium tank measures about two feet (60 cm) high. Yet it can fill over 50 balloons! How can such a small tank contain enough helium to fill so many balloons? The balloons don’t need a lot of helium, a small container can hold a lot of helium. Gizmo Warm-up The Boyle’s Law and Charles’ Law Gizmo™ shows a container of gas. In the container, the small purple spheres represent molecules. 1. Observe the particles. Are they all moving at the same speed? No, some move faster and others move slower 2. How do the particles interact with the walls and lid of the container? They bounce off the walls and lid of the container. These interactions contribute to the pressure on the walls of the container. Pressure is defined as force per unit area. The SI units of pressure are newtons per square meter (N/m2 ), or pascals (Pa). 3. Slowly drag the temperature (T) slider back and forth. (Note: In this Gizmo, the Kelvin scale is used to measure temperature. On the Kelvin scale, 0 degrees is absolute zero, the coldest possible temperature. Absolute zero is equal to -273.15 °C or -459.67 °F) A. How does the change in temperature affect the speed of the molecules? Higher temperature, faster speed. Lower temperature, slower speed. B. How does the change in temperature affect the volume of the container? Higher temperature, higher volume. Lower temperature, less volume. Activity A: Boyle’s law Get the Gizmo ready: Set the temperature (T) to 300 K. Check that the mass (m) is set to 0 kg. Question: How does pressure affect the volume of a gas? 1. Form hypothesis: In this experiment, you will pile weights on the lid of the container of gas. What do you think will happen as more weight is added to the lid? The volume will be less and the speed will be slower. 2. Notice: Look at the DESCRIPTION pane. What is the mass of the lid? 10 kg How much pressure does the lid exert on the gas? 98.1 N/m2 3. Collect data: With the temperature held constant at 300 K, use the Select mass slider to place weights on the lid. Record the pressure and volume of the gas for each added mass. Added mass on the lid Total mass (lid + added mass) Pressure* Volume 0 kg 10 kg 10 kg 20 kg 20 kg 30 kg 30 kg 40 kg *This model does not include atmospheric pressure, which is 101,325 N/m2 . 4. Analyze: As the pressure increases at constant temperature, what happens to the volume of the gas? This relationship is called Boyle’s law. 5. Calculate: Compare the pressure and volume values in your data table. A. How did doubling the pressure change the gas volume? B. How did tripling the pressure change the gas volume? C. How did quadrupling the pressure change the gas volume? (Activity A continued on next page) Activity A (continued from previous page) 6. Predict: If the added mass on the lid was 50 kg, a total mass of 60 kg would exert pressure on the gas inside the container. What will be the volume of the gas? 7. Test: Test your prediction using the Gizmo. What is the volume of the gas? Was your prediction correct? 8. Create a graph: On the GRAPH [Show More]
Last updated: 1 year ago
Preview 1 out of 12 pages
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Apr 16, 2022
Number of pages
12
Written in
Additional information
This document has been written for:
Uploaded
Apr 16, 2022
Downloads
0
Views
115








.png)




.png)
.png)








.png)
.png)

