IVF Surgery Exam1 Latest Graded A+
Document Content and Description Below
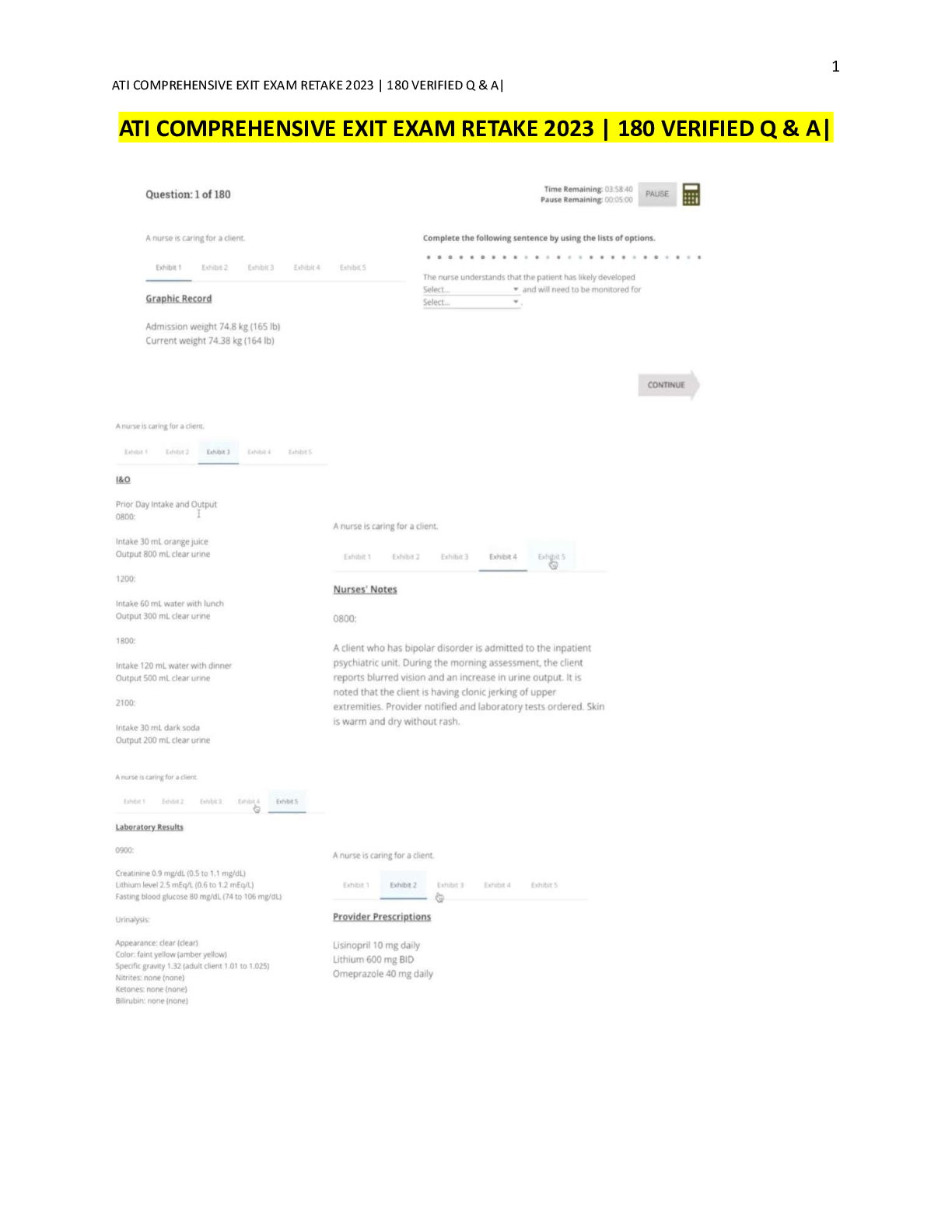
Total body fluid weight makes up ___% of total body weight? - ANSWER 60 2/3s of the body's volume is ___________ (intra/extracellular) - ANSWER Intracellular What is extracellular fluid comprise... d of - ANSWER Interstitial Intravascular 3rd space Daily water input? - ANSWER Food-800 Ingested- 500 Oxidation - 300 TOTAL-1600 mL Daily water output? - ANSWER Skin-500 Urine-500 Resp-400 Stool-200 TOTAL- 1600mL (35mL/kg/day or 2.4L) What instances do we need more water - ANSWER Burners? GI loss (V/D) Sweating Drains Tachypnea Polyuria Fever What is the solute concentration in the body fluid by weight? - ANSWER Osmolality NL- 275-295 3 types of IVF? - ANSWER Colloids Crystalloids Blood products These are fluids that have large molecules and do not pass cell membrane. Remain *intravascular* and draw *in* fluids from *high* oncotic pressure - ANSWER Colloids Fluids that contain small molecules that increase fluid in *interstitial* and *extravascular* spaces - ANSWER Crystalloids Three types of crystalloids - ANSWER Isotonic (MC) Hypertonic Hypotonic This is when the concentration of solutes is equal to that of plasma. Will this increase intra or extravascular volume? - ANSWER Isotonic crystalloids Increases intravascular volume (stays extracellularly) What is normal saline used to treat? - ANSWER Any hypovolemia Shock Mild hyponatremia Metabolic acidosis (DKA) What is the only fluid to be used with blood products? - ANSWER NS This is considered a balanced fluid, and the electrolyte content is most like that of blood serum and plasma - ANSWER LR Why does NS need to be used carefully in HF and RF? - ANSWER Replaces extracellular fluid and may cause overload What is contained in NS? - ANSWER Water Na (154) Cl (154) Similar to Na and Cl in intravascular space What is contained in LRs? - ANSWER Na (130) HCO3 (28) Cl (109) K (4) Ca (3) What are LRs used to treat - ANSWER GI loss Burns/trauma fluid loss Fistula drainage Acute blood loss or hypovolemia due to 3rd pacing Why are trauma fluid losses better treated the LR than NS? - ANSWER Electrolyte presence What does LR convert in the liver? What does this means in regards to acidosis - ANSWER Converts lactate to bicarb in liver Good for metabolic acidosis, *NOT* lactic acidosis What patients should you avoid LR with? - ANSWER NOT for liver pts (cant metabolize lactate) Caution in severe renal pt (K) Avoid in pts with pH >7.5 (bicarb) Is D5W hyper/hypo/isotonic? - ANSWER Considered isotonic, but when metabolized, is *hypo*tonic and causes fluid to shift into cells When is D5W indicated - ANSWER Hypernatremia Provides 170 calories/L but no electrolytes Cautionswith D5W? - ANSWER 1. ICP (cerebral edema) 2. HF/RF (overload) 3. Dilutes electrolytes (not monotherapy for hypovolemia) 4. Resuscitation (doesnt remain intravascaular) 5. Not immediate post op (increased ADH) What should be monitored with isotonic fluids - ANSWER -volume overload: look at vitals, volume status (JVD/edema), breath sounds (crackles), heart sounds All before, during and after -also I/Os-Are they peeing? These solutions often lower the serum osmolality within the vascular space, causing fluid to shift from intravascular space to both intracellular and intersitital spaces - ANSWER Hypotonic Hypotonic solutions hydrate the cells, but may also deplete fluid where - ANSWER Circulatory system Examples of hypotonic solution - ANSWER 1/2 NS 0.33% NS 0.22% NS 2.5% Dextrose in water When are hypotonic solutions used - ANSWER Intracellular dehydration -hypernatremia -DKA -HHS [Show More]
Last updated: 1 year ago
Preview 1 out of 8 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Aug 19, 2022
Number of pages
8
Written in
Additional information
This document has been written for:
Uploaded
Aug 19, 2022
Downloads
0
Views
39