Pharmacology > QUESTIONS & ANSWERS > Pharmacy Law MPJE Exam: Federal Law Questions and Answers 100% Pass (All)
Pharmacy Law MPJE Exam: Federal Law Questions and Answers 100% Pass
Document Content and Description Below
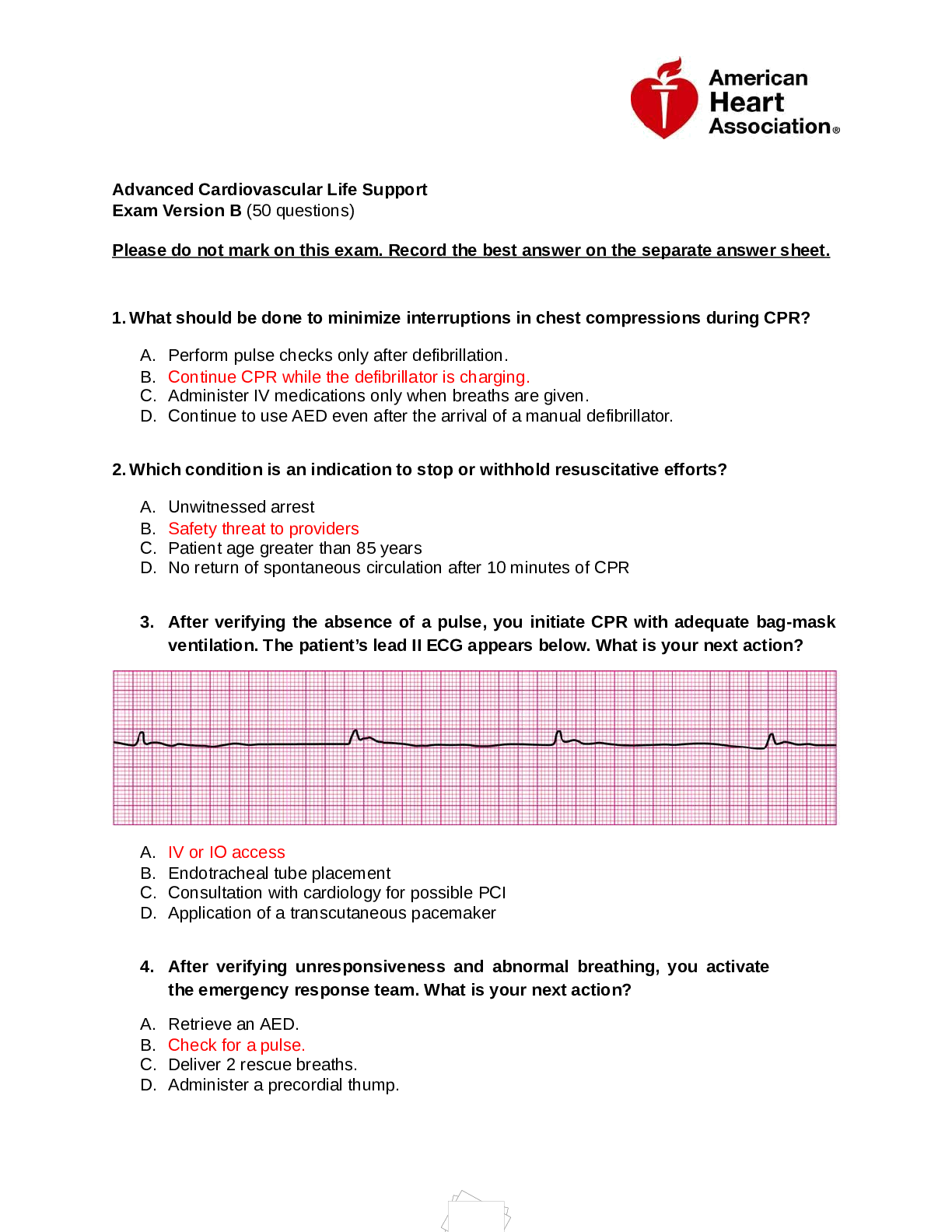
Pharmacy Law MPJE Exam: Federal Law Questions and Answers 100% Pass What did the Federal Controlled Substances Act do? Why was it implemented? ✔✔Legislators very concerned in the 1960s about le... gal and illegal drugs being abused They were given the opportunity to design an act/law Created the DEA to promulgate and enforce the law Created a closed system between: manufacturer, distributors, prescriber, dispenser, and patient. What did the Pure Food and Drug Act of 1906 do? ✔✔Prohibited the adulteration and misbranding of foods and drugs in interstate commerce; DID NOT have to be proven safe or effective; did not require the label to list ingredients, directions for use, or provide warnings What did the Food, Drug, and Cosmetic Act do? (started in 1938) ✔✔Provides for the comprehensive regulation of all drugs introduced into interstate (and most intrastate) commerce Under this act no new drug may be marketed and sold unless it has been proved SAFE ONLY for its intended use and approved by the FDA Labels must contain adequate directions for use and warnings about habit-forming properties of certain drugs Describe the FDCA ✔✔The purpose of the FDCA is to protect consumers from adulterated or misbranded foods, drugs, cosmetics, or devices; provided that no new drug could be marketed until proven safe for use under the conditions described on the label and approved by the FDA; also started the labeling requirements; applies to devices as well; all drugs before 1938 are exempt from the requirement that new drugs be proven safe If you get in trouble with the board of pharmacy can you get in trouble with the government too? What about the a lawsuit with a patient? ✔✔Yes, you can have an administrative lawsuit, a civil lawsuit, and a criminal lawsuit separately for the same offense What did the Durham-Humphrey Amendment of 1951 do? ✔✔Established 2 classes of drugs; prescription and OTC Allows refills on prescriptions and oral prescriptions Per the Durham-Humphrey Amendment; what must be labeled on a prescription medication ✔✔Caution: federal law prohibits dispensing without a Rx; they do not need adequate directions for use like OTC (talking about the MANUFACTURERS label here; there is no take this medication x amount of times daily on the manufacturer labeling) What did the Kefauver-Harris Amendment of 1962 do? ✔✔ [Show More]
Last updated: 1 year ago
Preview 1 out of 51 pages
.png)
Also available in bundle (1)
.png)
MPJE BUNDLED EXAMS QUESTIONS AND ANSWERS WITH VERIFIED SOLUTIONS
MPJE BUNDLED EXAMS QUESTIONS AND ANSWERS WITH VERIFIED SOLUTIONS
By Nutmegs 1 year ago
$27
23
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Mar 21, 2023
Number of pages
51
Written in
Additional information
This document has been written for:
Uploaded
Mar 21, 2023
Downloads
0
Views
76

.png)
.png)
.png)
.png)
.png)
.png)








.png)
.png)
.png)
.png)
.png)

.png)
.png)
.png)
.png)
.png)




